If you search the internet for information on the relationship between plant foods and gout, uric acid, urea or purines, you are very likely to come across a wide range of viewpoints – from those saying that you should avoid purine-containing plant foods and that they therefore increase gout symptoms, to those saying that plant foods (including those high in purines) can actually reduce gout symptoms. But what does the research say?
However, let’s first look at what gout is and what uric acid, urea and purines have to do with it.
Blog Contents
What Is Gout?
The Arthritis Foundation 1 gives the following information about gout.
- Gout is a form of inflammatory arthritis that develops in some people who have high levels of uric acid in the blood
- The acid can form needle-like crystals in a joint and cause sudden, severe episodes of pain, tenderness, redness, warmth and swelling
- Gout can be broken down into four stages:
- Asymptomatic hyperuricemia – period prior to the first gout attack
- No symptoms
- High blood uric acid levels
- Crystals starting to form in the joint/s
- Acute gout (also known as gout attack) – Something causes uric acid levels to spike or jostles the crystals. For instance, a night’s heavy drinking or physically knocking a joint against a hard object. This spike or the movement of crystals in the joint results in:
- Inflammation and pain
- Attacks usually strike at night
- Intensifies over the next 8-12 hours
- Symptoms ease after a few days
- Symptoms likely to disappear in 7-10 days
- A second attack is not always experienced – ~60% of people have a second attack within 12 months. ~84% have another attack within three years
- Interval gout – the time between attacks
- No pain, but the gout isn’t gone
- Low-level inflammation may still be damaging joints
- This is usually when doctors try to get patients to manage their gout to prevent chronic gout, via:
- Medication (see below)
- Lifestyle changes (diet, alcohol intake, exercise, etc)
- Chronic gout – develops when high uric acid levels remain untreated over a number of years.
- Attacks become more frequent
- Pain may not go away as it used to
- Joint damage may occur
- Mobility may be lost
- Asymptomatic hyperuricemia – period prior to the first gout attack
The Risk Factors for Gout
- Genes: Increased risk if family member/s have gout – although this is still not well understood
- Health conditions that may increase risk:
- High cholesterol
- High blood pressure
- Diabetes
- Heart disease
- End-stage renal disease
- Medications: (See Appendix 1. for detailed information on medications that are prescribed for gout.)
- Gender and age:
- More common in men as they age, but then common in post-menopausal women (oestrogen may protect women up to that point)
- Diet: [see later for more information on dietary factors]
- Red meat
- Shellfish
- Alcohol: More than 2 measures of spirits/pints of beer a day can increase the risk
- Sodas/Soft Drinks: Risk is increased probably because of the high levels of high fructose corn syrup
- Obesity:
- Generally linked to a higher risk
- Obese individuals tend to develop it at a younger age than people of normal weight
- Bypass surgery: Gastric bypass surgery increases risk
- Lead contamination6
Uric Acid, Urea & Purines – What are They & How Do They Relate To Gout?
- Urea is the main breakdown product of protein catabolism in mammal cells and is excreted in urine
- Uric acid is the main breakdown product (metabolite) of purine metabolism in mammals, it is produced in the liver and is also excreted in urine
- Purines can be regarded as one of the building blocks of all living things, being compounds made of carbon and nitrogen atoms that are in the nucleus of every cell within all living plants and animals
Urea & Uric Acid – Waste Products
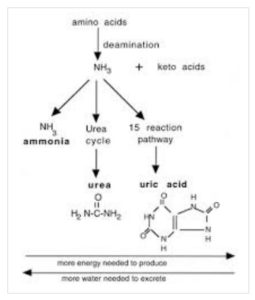
The human body produces a lot of unnecessary and toxic by-products during metabolism (the breakdown of compounds in food to produce the energy we need to live). Urea, uric acid and also ammonia are nitrogenous excretory products which are safely removed from a healthy body by first being converted to less toxic substances, and then excreted as urine and, to a lesser extent, as sweat (along with sodium chloride – salt – with the skin acting as the excretory organ).
Regarding urine, the main excretory organs are the kidneys. These produce urine, and this is the primary method of excreting the excess and unnecessary substances from our bodies, as part of the process called osmoregulation which controls the water/salt concentrations within the body. (See Appendix 3. below for a detailed look at osmoregulation and how urine is formed in the kidneys.)
Ammonia is a by-product of protein metabolism and is highly toxic and needs to be removed from the body. However, before it can be removed, it needs to be converted to something less toxic – namely, urea.
Urea has the molecular formula CO(NH2)2 . It’s naturally produced in mammals in the metabolism of nitrogen (found in protein within the foods we eat) in the urea cycle which is a vital metabolic process 7 . When ammonia or amino acids are oxidised inside our bodies, urea is produced as a colourless, odourless non-toxic solid which is then highly soluble in water – very helpful when excreting it from the body. Urea of itself is not the direct problem for gout, so long as the kidneys are functioning well – that is, removing urea from the blood and converting it to urine for safe excretion. 
Uric acid is regarded as the gouty ‘bad boy’ and is a heterocyclic compound of carbon, nitrogen, oxygen, and hydrogen with the formula C₅H₄N₄O₃. It forms ions and salts known as urates and acid urates, such as ammonium acid urate. Like urea, uric acid is a crystalline solid but, unlike urea, its water solubility is generally much lower. If we were birds or reptiles, we would excrete uric acid as a solid, but we have to dissolve it in our blood and then pass it into our kidneys to be either reabsorbed or urinated out of the body. If we can’t dissolve and excrete enough uric acid, it can result in crystals travelling to joints and causing excruciating pain. if elevated uric acid levels continue, kidney stones may end up being formed – not something anyone wants to have.
Normal levels of urea & uric acid in blood tests
The definition of what is a “normal level” varies from laboratory to laboratory. The laboratory I use for my blood tests uses the following levels:
- Normal urea levels are between 1.70 and 8.30 mmol/L
- Normal uric acid levels are between 266.00 and 474.00 umol/L (4.47 mg/dL* – 7.97 mg/dL)
* dL or decilitre is 1/10 or 10% of a litre. So, if a litre is 1000 ml, a decilitre (dL) is 100 ml .
Internet tools for converting uric acid measurements (mmol or umol/L to mg/dL) are available.8 As well as those for converting mmol (millimoles) to umol (micromoles). 9
Whatever your laboratory defines as the normal levels, if urea blood levels are higher than normal (perhaps caused by too much dietary protein, dehydration or impaired kidney function), it is likely that there will also be an excess of uric acid, too. If your blood test test shows elevated uric acid blood levels, it’s not just gout that might be your problem. Elevated uric acids levels also have associations with diabetes, cardiovascular disease and kidney stones.
Conversely, low uric acid blood levels can be associated with MS (multiple sclerosis).
Gout Podcast by Dr Joseph Lieberman 10
This highly informative podcast by Dr Joseph Lieberman11 , Professor of Family Medicine at Jefferson Medical College in Philadelphia delves deeper into how the current understanding of gout is changing.
The following is a summary 12 of points discussed:
- Laboratory definitions of “normal” serum uric acid levels (6.0-9.0 mg/dL / 356.88-535.32 umol/L in the USA) are not the same as clinical definitions of clinically significant levels for individual health. Laboratories take the average (normal) levels of a population range, while clinicians are concerned about the lowest level at which uric acid concentration poses a threat
- The deposition of the crystals (monosodium urate crystals) into soft body tissues occurs when the the serum urate level is reaches 6.8 mg/dL (404.46 umol/L). Above this solubility level, the blood can no longer hold the uric acid in liquid form and so it precipitates out into crystals. Once the crystals are in the blood, they can then travel into soft tissues and eventually into the space between joints, causing the well-known painful symptoms
- Hyperuricaemia (an excess of uric acid in the blood) in the absence of crystal deposition in the soft tissues is not classed as gout. Therefore, Dr Lieberman considers that asymptomatic (lacking any symptoms) hyperuricaemia generally does not require any treatment. However, there should be a cause for concern that serum uric acid levels are higher than 6.8 mg/dL (404.46 umol/L) and some lifestyle changes would be highly recommended (see below)
- If the serum urate acid level is above 8.0 mg/dL (475.84 umol/L), this is certainly high enough to get concerned and clinical advice should be sought. At this level, it is pretty much certain that crystal deposition is occurring, even in the absence of any symptoms at the time of testing
- Dr Lieberman recommends that serum uric acid levels should be kept below 6.0 mg/dL (356.88 umol/L) in order to reduce the risk of precipitation of crystals into soft tissue
- Under-excretion of uric acid accounts for around 90% of cases of hyperuricemia while overproduction of uric acid accounts for around 10% [Purines can be endogenous (manufactured by your own body) or exogenous (entering the body via food)]
- For gout to develop, an elevated serum urate level along with local factors, such as increased temperature and/or trauma, must be present. Once the monosodium urate crystals are in the joint spaces, they initiate an acute inflammatory response. This can be self-limiting and, though painful when occurring, it may only last for between 3 days and two weeks and then disappears
- However, chronic crystal deposition can lead to low-grade inflammation which eventually damages joints.
- Serum urate acid levels are not the definitive test for gout. That is, you can have normal levels but still have an acute attack of gout because of the crystals already in the spaces between the joints. If you do have a high urate acid level, though, this is a strong indicator that you could be on route for a gout attack, certainly if the level is above the 6.8 mg/dL (404.46 umol/L) distillation point
The “perfect storm” for gout incidence
Specifically in the USA, but applicable to all developed countries, Dr Lieberman considers that we now have a “perfect storm” for an increase in the prevalence of gout. Contributing factors include:
- Increased longevity of the population (although not increased length of disease-free, fully-healthy years)
- Increased prevalence of lifestyle diseases:
- Iatrogenic causes (effects of medical treatments themselves) such as increased use of cyclosporine and thiazide diuretics
- Dietary trends, including the widespread presence of high-fructose corn syrup in many prepared foods [Interestingly, Dr Lieberman does not mention the problem of the increase in consumption of animal protein over the past decades]
- Limited therapeutic armamentarium (the medicines, equipment, and techniques available to a medical practitioner)
Diuretics and Gout
Dr April Chang-Miller wrote the following18
Yes. Diuretics can increase your risk of developing gout, a type of arthritis caused by the buildup of uric acid crystals in a joint.
This may happen because diuretics increase urination, which reduces the amount of fluid in your body. But the remaining fluid is more concentrated, which can increase the risk that you’ll develop the crystals that cause gout. Some types of diuretics also reduce the kidneys’ excretion of urate, a component of uric acid.
You and your doctor will decide if it’s best for you to continue taking the diuretic, or switch to another medication. There are many other types of blood pressure medications that don’t increase your risk of gout. Also, many of the measures you take to reduce blood pressure have the added benefit of lowering uric acid. Other measures that you can take to lower uric acid include:
- Eating a healthy diet, with an emphasis on vegetables, plant proteins, low-fat dairy products and whole grains, and reduced servings of meat and seafood
- Drinking little to no alcohol
- Limiting soft drinks/soda beverages that are sugar sweetened and limiting foods and beverages that contain high fructose corn syrup
- Losing weight and maintaining a healthy weight based on your body mass index (BMI)
- Stay hydrated by drinking plenty of water
- Limit intake of purine-rich foods. Common purine-rich foods include liver, mackerel, herring, game meats and sardines
So how do purines fit into this?
Eating purine-high foods is thought to increase the amount of purines in the blood and hence increase the amount of uric acid as the purines are broken down.
Foods that are high in purines and are conventionally considered to represent a potential problem for gout include:
- Animal foods
- Organ meats (e.g. liver)
- Sweetbreads (e.g. thymus gland)
- Beef
- Poultry
- Pork
- Fish (e.g. anchovies, sardines, mackerel and scallops)
- Plant foods
- Asparagus
- Cauliflower
- Dried peas & beans
- Lentils
- Mushrooms
- Spinach
- Oatmeal
- Wheat bran/germ
- Yeast
- Alcoholic drinks (particularly beer)
- High fructose corn syrup (and fructose itself)
- Salt (including sea salt)
Some people have a problem metabolising purines, such as people with hyperuricemia or gout, and they are advised to limit consumption of the above foods. However, evidence suggests that it is the purines in animal foods, and not in plant foods, that increase gout risk.
Consumption of purine-rich vegetables was not associated with gout according to a 2005 study19 . The same study considered that total protein was not a problem and dairy consumption was helpful for gout. It concluded: “These findings from a nationally representative sample of adults in the US suggest that higher levels of meat and seafood consumption are associated with higher serum levels of uric acid but that total protein intake is not. Dairy consumption was inversely associated with the serum uric acid level.”
A 2009 prospective study20 concluded: “Higher vitamin C intake is independently associated with a lower risk of gout. Supplemental vitamin C intake may be beneficial in the prevention of gout.”
Interestingly, a 12-year prospective study 21 found that “…long-term coffee consumption is associated with a lower risk of incident gout.” The researchers added: “Total caffeine from all sources and tea intake were not associated with the risk of gout.”
A 2012 case-crossover study22 concluded that: “…acute purine intake increases the risk of recurrent gout attacks by almost five times among patients with gout. The impact from animal purine sources was substantially greater than that from plant purine sources. Avoiding or reducing purine-rich foods intake, especially of animal origin, may help reduce the risk of recurrent gout attacks.”
A 2011 review stated that: “Based on the epidemiological studies …dietary modification to increase regular low fat milk or yoghurt, vegetable sources of protein, cherries and coffee, and to decrease intake of liver, kidneys, shellfish, seafood, yeast extracts, total protein, and red meat would appear reasonable.” This was endorsing the findings of a 2007 BSR & BHPR executive summary23
Gout Prevention Diets
If we accept that diet is a leading cause of gout – since most of the above non-communicable lifestyle diseases listed above are linked to what we do or don’t put in our mouths – then what is the best diet for gout prevention?
The following is from Arthritis-Health24 :
| High Purine Foods | Moderate Purine Foods: Eat Limited Quantities | Low Purine Foods |
| Meats, especially organ meats or “sweetmeats,” such as liver, brains, and beef kidneys, as well game meats, such as venison, which are typically fatty | Certain vegetables, including asparagus, spinach, mushrooms, green peas and cauliflower (no more than ½ cup per day) | Any vegetables that are not listed as moderately high in purines, such as leafy greens, carrots and tomatoes |
| Foods containing saturated fats: these tend to inhibit the body’s ability to metabolize purines | Beef, pork, lamb, fish and poultry (no more than 4-6 oz daily) | Condiments that contain oils, spices, and vinegars are generally acceptable |
| Seafood, particularly scallops and other shellfish, anchovies, sardines, herring, and mackerel | Wine* (1-2 glasses, when gout symptoms are absent) | Rice, enriched pastas and breads, potatoes, and popcorn |
| Foods and drinks made with high fructose corn syrup, such as sodas25 | Wheat bran and wheat germ (1/4 cup dry daily) | Nuts and nut products, such as peanut butter |
| Supplements containing yeast or yeast extract | Dried beans, lentils and peas (1 cup cooked) | Dairy products (preferably low- or no-fat) |
| Gravy | Oatmeal (2/3 cup dry daily) | Eggs, particularly egg whites |
| Beer* | Fruit juice (no corn syrup) | Coffee and tea |
| Meat-based soup stocks | Fruits |
*Alcoholic drinks can inhibit the body’s ability to eliminate uric acid, so people with gout are advised to avoid alcohol or drink in moderation. Beer is notorious for bringing on gout attacks because it contains both alcohol and brewers yeast, which is high in purines. Nutritional/brewer’s yeast is also something that should be avoided by those who have Crohn’s disease.26
People on a low-purine diet should drink plenty of water to aid with digestion and lower uric acid concentrations in the blood.
Writers for the Arthritis-Health website continue with the following suggestions for a gout prevention diet27 :
- Rather than completely avoiding, one should merely limit meat and seafood consumption to no more than 4-6 ounces of meat, poultry or seafood per day if you have gout
- Plant-based proteins (e.g. vegetables, nuts and legumes) and complex carbohydrates
- Replacing some meat and seafood with legume consumption (fresh, canned or frozen, not dried) will decrease purine intake
- High-protein diets should be avoided
- Gout sufferers also will benefit from avoiding foods made with sugar and high fructose corn syrup (HFCS) and sodas25 . Instead, complex carbohydrates, such as fruit and whole-grain products, are better
- Low-fat dairy products should be chosen over full-fat. The justification of this derives from the 2005 Third National Health and Nutrition Examination Survey19 which expressed the opinion that people who consume low-fat dairy products such as skimmed milk and yogurt can decrease their levels of uric acid and thereby decrease their risk of gout attacks. The Arthritis-Health website cites supporting opinion from the Mayo Clinic28 , although it appears that the Mayo Clinic merely refers back to the latter-mentioned survey when they recommend 16 to 24 ounces of daily low-fat dairy consumption per day, pointing out that high fat dairy products don’t have the same protective effect
- Many people believe eating cherries or drinking cherry juice will help fend off gout, and there is some scientific evidence to support this.29 30 (See below for Dr Greger’s comments on cherries and gout-prevention/treatment)
- Some otherwise healthy plant foods are also high in purines and should be eaten in moderation:
- Asparagus
- Spinach
- Mushrooms
- Dried beans
- Peas
- Avoid alcohol
- Maintain a healthy weight
Dr Michael Greger’s Viewpoint
The following video31 by Dr Greger, “Gout Treatment with a Cherry on Top” (followed by an abbreviated transcript, and also available as a separate nutritionfacts.org article32 ), presents a clearly positive view about the role of cherries in treating gout.
Abbreviated transcript of the above video:
- It’s known that cherries are anti-inflammatory in research with rats and humans.33 34 35 36 . Reductions in CRP (C-reactive protein – a common marker for inflammatory responses in the body) even continued a month after the study ended in both healthy and less healthy subjects
- But what about gout?
- Research as far back as 195037 showed that gout flare-ups in gout patients were helped by “eating [half a pound] of fresh or canned cherries”
- In a 2012 study38 , “[c]herry intake…was associated with a 35% lower risk of gout attacks,” with only three serving already reducing the risk by half. So around 16 cherries a day had the same effect as a low-purine diet
- The same research team extended the latter 2012 study39 , finding that:
- “[P]urine intake of animal origin increased [the odds for recurrent] gout attacks by nearly fivefold.”
- Heavy alcohol consumption was a bad idea
- High-purine plant foods, like mushrooms and asparagus, had no significant link to the risk of developing gout
- The best recommendation is to eliminate meat and seafood from the diet but add cherries
- eating cherries combined with taking the leading gout drug, allopurinol works even better
- So should gout sufferers ask their doctor for uric acid-lowering drugs, like allopurinol?
- This is probably not such a good idea and, in any case, doctors don’t like to prescribe uric acid-lowering drugs because of “rare but serious side effects”— including the most feared of all drug side effects: Stevens-Johnson syndrome, which can detach your skin from your body. Here is a link to what it can look like – but only look at this if you have a strong stomach! [Not worth the risk in my opinion]
- Research mentioned earlier 40 has shown that not only do they fight inflammation, but cherries may actually help lower uric acid levels, as well. “Within five hours of eating a big bowl of cherries, uric acid levels in the blood significantly drop. At the same time, antioxidant levels in the blood go up, as vitamin C levels start to rise.“
- Do other fruits have the same effect? We know that fruits in general have antioxidant effects; but what about being able to reduce serum uric acid levels? The answer was no. Grapes, strawberries, and kiwi fruit were tried and none lowered uric acid levels to a significant level. This supports the idea that cherries have a specific “anti-gout effect”
- New (very expensive) gout drugs are being developed, but some researchers consider41 that these drugs carry “a risk of toxicity that may be avoided by using nonpharmacologic treatments or [prevention]” by making simple dietary modifications, including reducing alcohol and animal food intake, and eating cherries
An additional article by Lori42 , a contributor to nutritionfacts.org, adds:
- [Nutritional/brewers] Yeast (a natural immune system regulator) is high in purines, so those with gout may want to limit consumption to less than a teaspoon a day
- Higher consumption of purine-rich plant foods like cauliflower, mushrooms, peas, beans, asparagus, and lentils appear to be protective. Contrary to conventional medical advice, eating purine-rich plant foods have not been associated with high uric acid levels. “This may be because foods rich in fiber, folate, and vitamin C appear to protect against uric acid buildup and gout. For example, fiber has been recognized as having a potential role in binding uric acid in the gut for excretion.”43
- Cutting out all soft drinks/soda beverages may also reduce the risk of gout
- In general, eating a more alkaline diet [which naturally happens on a no-SOS WFPB diet] has been found to flush significantly more uric acid from the body, decreasing the uric acid levels in the blood. In an 2003 interventional trial44 it was found that, in only a few days, participants experienced a 93% decline in the risk of uric acid crystallisation. [N.B. This trial only used a vegetarian diet and not the optimal WFPBD. More 3rd party information on alkaline diets and gout is available online 45 ]
- It has been claimed46 that Coriander (cilantro) is also effective in reducing uric acid levels. This is supported by a 2012 study47 , which concluded that: “Coriander leaves significantly influenced almost all the parameters in arthritis patients without any detrimental effects by virtue of a number of phytochemicals, vitamins and minerals present in the leaves having therapeutic effects. The antioxidant and antiarthritic activities exhibited by the leaves are a result of the synergistic action of the bioactive compounds present in the leaves.”
Joe’s Comments
The foregoing is just a snapshot of the mass of information and opinion available on gout causes and cures. It seems intuitively likely that an alkaline diet, high in fibre and antioxidant/antiinflammatory plant foods would be the ideal diet for the prevention of high serum uric acid levels and for the treatment of existing gout flare-ups. The evidence seems to point clearly towards animal foods and alcohol being major causal factors in the incidence of gout symptoms.
For my part, all my uric acid levels have been very low in blood tests since I started eating a WFPBD (3.4 – 4.4 mg/dL / 202 – 261 umol/L). This is, of course, merely one case study.
The research on low-fat milk was interesting. There are so many other reasons not to consume any dairy products48 , so does the above research mean people worried about getting gout who already eat (or are thinking of eating) a WFPB diet should add cow’s milk to their daily intake? I would hesitate before doing this for two reasons: firstly, the research that indicated the benefits of low-fat milk was comparing it with full-fat milk, and research has suggested for a long time (as far back as 193949 ) that dietary fat inhibits the ability of the body to excrete surplus uric acid; and, secondly, there has not been any research, to the best of my knowledge, on the dietary effects on uric acid levels when the two diets being compared are one including cow’s milk and the other including either plant milks and/or WFPB diets.
There appears to be no research showing negative effects on uric acid levels when plant foods (excluding nutritional/brewers yeast) are consumed. Indeed, research mentioned above supports the hypothesis that the very plant foods high in purines that are conventionally regarded as harmful are, in actual fact, protective against gout.
I will continue to update this article with new research as it is published. In the meantime, if you have any personal experiences relating to this topic that you would like to share, please write to me at [email protected].
Pseudogout
By the way, there is a similar condition called pseudogout which can be confused with gout since it shares some symptomatic similarities (see appendix 2. for more details).
Appendix 1. What Are The Usual Drug Treatments?50
The drugs used* to treat gout tend to be aimed at reducing pain, inflammation and uric acid levels in the blood. The following are some of the common medications with benefits and risks along with known side effects.
Colchicine (Colcrys)
Colchicine is an oral anti-inflammatory prescribed for acute attacks or when beginning uric acid-lowering drugs.
Benefits and risks: Colchicine treats the pain and inflammation of acute attacks. It is most effective if taken at the first signs of a gout attack. It is available along with probenecid, a drug that helps the body eliminate uric acid, in a single pill under the trade name Col-Benemid.
It prevents gout flares when starting a medicine that lowers uric acid levels. You should not take colchicine if you have liver or kidney disease.
Side effects: Common side effects include diarrhoea, nausea and abdominal cramps.
Corticosteroids
Corticosteroids are powerful anti-inflammatory drugs that may be prescribed to control pain and inflammation of acute attacks.
Benefits and risks: Corticosteroids work rapidly to reduce inflammation and can be taken orally, injected systemically or directly into inflamed joints. They can cause serious health problems if used in high doses or for a long time. Frequent joint injections could lead to ligament and cartilage damage.
Injection of adrenocorticotropic hormone (ACTH), a synthetic drug that stimulates the body to produce corticosteroids naturally, can also help treat a gout attack.
Side effects: Common side effects include increased appetite and weight gain, easy bruising, increased body hair and decreased resistance to infection.
Nonsteroidal anti-inflammatory drugs (NSAID’s)
NSAID’s are a large class of drugs used to relieve pain and inflammation. In gout, they are often given during acute attacks.
NSAID’s may be prescribed along with a uric acid-lowering drug for the first 6 weeks to 12 months to prevent attacks.
Benefits and risks: NSAID’s can help relieve the pain of an acute attack and shorten the attack, especially if taken in the first 24 hours. The most serious risks include stomach bleeding, kidney failure, and cardiovascular problems.
Side effects: Common side effects include nausea and vomiting, headache, diarrhoea, dizziness, rash and drowsiness.
Allopurinol (Zyloprim)
Allpurinol is an oral medicine called an xanthine oxidase inhibitor (XOI), which reduces the body’s production of uric acid.
Benefits and risks: Allopurinol reduces how often gout attacks occur and makes them less severe. However, you may be at increased risk of an attack immediately after starting this drug. Taking allopurinol during an attack may actually make it worse. In rare cases, it can cause a severe allergic reaction.
Allopurinol is often prescribed at a low daily dose at first, with the dosage gradually increasing over time. It is also available along with lesinurad, another gout medication, in a single pill under the brand name Duzallo.
Side effects: Occasional side effects include skin rash and stomach upset. Stomach problems usually go away as your body adjusts to the drug.
Febuxostat (Uloric)
Febuxostat is an oral XOI that reduces the body’s production of uric acid.
If your gout flares while taking febuxostat, do not stop taking the drug. Contact your doctor. Don’t take febuxostat if you are taking azathioprine or mercaptopurine 51 .
Benefits and risks: Febuxostat can reduce how often attacks occur and make them less severe. You may experience a flare when you first start taking the drug. In rare cases, serious skin rashes occur.
Side effects: Common side effects include nausea, joint pain and muscle pain.
Lesinurad (Zurampic)
Lesinurad is an oral drug that helps the body eliminate uric acid. It is prescribed along with an XOI, such as allopurinol or febuxostat.
Stay well hydrated while taking lesinurad to avoid kidney stones. Lesinurad is also available with allopurinol in a single pill under the trade name Duzallo.
Benefits and risks: Lesinurad enhances the effects of XOIs to help control uric acid levels when an XOI alone is not sufficient. However, it may also increase cardiovascular risk.
Side effects: Possible side effects include headache, flu-like symptoms, increased blood creatinine, gastroesoephageal reflux disease, kidney-related side effects and kidney stones.
Pegloticase (Krystexxa)
Pegloticase is given by intravenous infusion. It changes uric acid into a substance called allantoin that your body can eliminate easily. It is reserved for people in whom other gout medications have not worked.
Pegloticase is administered every two weeks at an infusion centre. Your doctor may prescribe medicines before the infusion to reduce the risk of a negative reaction. You should not use any other drugs to lower uric acid while you are receiving pegloticase.
Benefits and risks: Pegloticase is often effective in controlling gout when no other drugs do. Rarely, serious allergic reactions occur, usually within two hours of the infusion.
Side effects: Common side effects include gout flares or attacks, allergic reactions, nausea, bruising, sore throat, constipation, chest pain and vomiting.
Probenecid (Generic)
Probenecid is an oral drug that helps your kidneys eliminate uric acid.
Probenecid is taken daily, even when you are not having symptoms. You should stay well hydrated to reduce the risk of kidney stones. Probenecid is also available along with colchicine in a single pill under the name Col-Benemid.
Benefits and risks: Probenecid reduces gout attacks and their severity. However, it may increase the frequency of attacks the first 6 to 12 months you take it.
Side effects: Common side effects include kidney stones, nausea, skin rash, stomach upset and headaches.
*By listing these drugs, the writer does not aim to give any medical advice or suggestions regarding their use. They are listed purely for academic reasons. If you consider you require medication, seek professional advice from you doctor.
Appendix 2. Pseudogout52 53
What is pseudogout?
Pseudogout, also known as calcium pyrophosphate deposition (CPPD) disease, is a type of arthritis that causes spontaneous, painful swelling in your joints. It occurs when crystals form in the synovial fluid (the fluid that lubricates the joints). This causes inflammation and pain. This condition most commonly affects the knees, but it can also affect the ankles, elbows, and wrists. The symptoms can last anywhere from a few days to several weeks. It’s more common in older adults. While you can’t prevent the disease, you can find treatments to reduce the inflammation and relieve the pain.
What are the causes of pseudogout?
Pseudogout occurs when calcium pyrophosphate crystals form in the synovial fluid in the joints. Crystals can also deposit in the cartilage, where they can cause damage. Buildup of crystals in the joint fluid results in swollen joints and acute pain. Researchers don’t fully understand why crystals form. Some contributing factors may include:
- Hypothyroidism (underactive thyroid)
- Haemochromatosis (also known as iron-storage disease where the body absorbs too much iron)
- Hypomagnesaemia (magnesium deficiency)
- Hyperparathyroidism (Overactive parathyroid gland)
- Hypercalcaemia (too much calcium in the blood)
- Genetic factors – recent research has found that an abnormality of the ANKH gene may lead to the production of too much pyrophosphate, which can result in widespread calcium crystal depositing. This can cause recurrent attacks of acute CPP crystal arthritis at an unusually young age (20s or 30s)
- Uncommon causes of calcific tendinitis (an associated condition) include:
- Diabetes
- Kidney malfunction
- Hypercalcaemia
What are the symptoms of pseudogout?
- Bouts of joint pain
- Swelling of the affected joint
- Fluid buildup around the joint
- Chronic arthritis
Is pseudogout the same as gout?
No it’s not the same thing, although the they share the fact that crystals – monosodium urate crystals in the case of gout, and calcium pyrophosphate crystals (CPPC) in the case of pseudogout – get into the spaces between joints and cause swelling, acute pain and, if untreated, can cause long-term damage. However, treatment for pseudogout with antiinflammatory medications is generally highly effective and only untreated and repeated incidences appear to lead to joint damage.
Calcium pyrophosphate crystals can damage tissue
Crystals are made up of very small geometric particles (atoms) arranged in a regular repeating pattern. This makes them very hard and difficult to break down. In certain substances this is helpful – for example, it contributes to the strength of bone and seashells. However, the hard sharp angles of calcium crystals can rub and grind down softer substances that come into contact with them. In addition, their rough surface has a strong electrical charge, which can injure cells and trigger an immune reaction (inflammation).
Even if you’re perfectly healthy, the chemicals that form crystals may be present in your blood, urine or soft tissues. Other chemicals within the body either promote or inhibit the formation of crystals, and the balance between these chemicals tends to change as part of normal ageing. These changes are mainly localised to particular parts of the body and are the most likely reason for crystals forming.
What triggers acute calcific crystal attacks?
Many attacks of acute CPP crystal arthritis and acute calcific tendinitis occur for no obvious reason, so it’s not clear why the crystals have been shed. But sometimes the cause of an acute attack will be more apparent, such as:
- An injury to your knee or shoulder may shake the crystals loose, setting off an attack a day or two afterwards
- An illness that causes a fever, such as having flu or a chest infection can, for some reason, also encourage crystal shedding
- A major stress to your body – such as having an operation or a heart attack
Appendix 3
Osmoregulation – How urine is formed in the kidneys and what role ADH plays
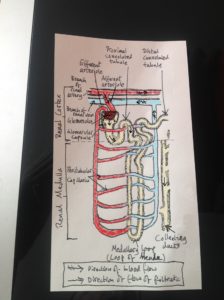
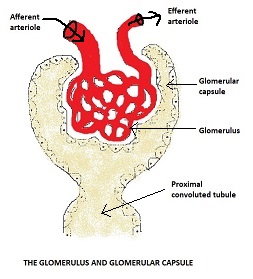
The kidney nephron produces urine by exchanging substances with the blood that arrives at the glomerulus from the blood capillaries which are supplied blood from the afferent arteriole which itself is supplied blood from the renal artery. Each kidney contains over one million nephrons and each has a glomerulus (the site of blood filtration), which is the diagrams show is a network of capillaries surrounded by a cup-like structure called the glomerular capsule (also known as the Bowman’s capsule).
The kidneys are thus able to filter unwanted substances from the blood into urine and excrete them. Many substances will, of course, remain in the bloodstream throughout this urine-production process without being absorbed into the glomerular capsule and will continue along the efferent arteriole and return to the circulatory system. Others will pass through the semipermeable membrane of the glomerulus and be reabsorbed in later stages of urine-production. Yet others, waste materials and some water, will continue until excreted from the body as urine.
The three main steps involved in this urine-production process ensure that only waste materials and excess water are removed from the body. These three steps in the formation of urine are: glomerular filtration, selective reabsorption and secretion.
Glomerular filtration starts the formation of urine. Some substances within the blood filter through the semipermeable membrane of the glomerulus and into the glomerular capsule. These substances include the small water (H20) molecules, although some water is then reabsorbed later in the process. Waste products, including those from protein metabolism, are also passed through the glomerulus membrane. At this stage there are two forms of pressure at work – osmotic and and hydrostatic. In the blood capillaries of the arterioles, hydrostatic pressure tends to increase filtration by pushing the latter small substances (also known as solute) and fluid (also known as solvent and primarily water at this stage) out of the capillaries and into the filtrate within the glomerular capsule. At the same time, osmotic pressure (also known as colloid osmotic pressure or oncotic pressure) pulls the fluid into the capillaries and/or prevents some fluid from leaving them. During this stage, electrolyte, water and pH levels are maintained – the latter via the excretion of hydrogen ions.
The resulting glomerular filtrate (that is, the substance that have been filtered) will typically contain mineral salts, ketoacids, amino acids, glucose, some hormones, creatinine, urea, uric acid, and the small molecules of some drugs. At the same time, the blood that remains in the glomerular capillaries will typically contain plasma proteins (e.g. serum albumin), erythrocytes, leukocytes, platelets, and the larger molecules of some drugs.
It will hopefully be seen from attached diagrams of the kidney nephron that the efferent arteriole is narrower than the afferent arteriole. A result of this is that a hydrostatic pressure within the capillary of around 7.3 kPa (55 mmHg) builds up within the glomerulus. The opposing osmotic pressure within the blood (mainly provided by the plasma proteins), is around 4 kPa (30 mmHg). The additional hydrostatic pressure within the filtrate of the glomerular capsule is around 2 kPa (15 mmHg). The resulting calculation of net filtration pressure can thus be seen as 7.3-(4+2)=1.3 kPa, or 55-(30+15)=10 mmHg.
The way that healthy kidney output is measured is by calculating the volume of filtrate produced (by both kidneys) each minute. This is called the glomerular filtration rate or GFR. A healthy adult has a GFR of about 125 ml/min. This equates to the formation of around 180 litres of filtrate a day (by both kidneys combined).
It is important to mention autoregulation, which is the mechanism that maintains a constant renal blood pressure even when systolic blood pressure is varying, for instance during daily activities (it would be fair to say that normal systolic variation across an average population would be something between 80 and 200 mmHg). The autoregulation mechanism thus maintains a more-or-less constant renal blood flow and, hence, constant level of glomerular filtration. Operating independently of nervous system control, renal autoregulation will continue unless there is an interruption in nerve supply to the renal blood vessels or if systolic blood pressure falls below around 80 mmHg. At this stage, autoregulation would fail and filtration within the glomeruli would be impaired by the drop in hydrostatic blood pressure within the capillaries.
Selective reabsorption is the next step of urine-production. Almost all the filtrate is reabsorbed from the kidney tubules with <1% actually being excreted as urine. This would be between 1 and 1.5 litres approximately per day. This selective reabsorption of most of the filtrate accounts for the differences in the total volume and eventual concentration of the urine. The majority of filtrate reabsorption occurs in the proximal convoluted tubule – “proximal” means “near” – (see attached diagram of kidney nephron – it is highlighted in a mustardy yellow colour). Microvilli (tiny hair-like projections) line the inner walls of this tubule in order to increase surface area and hence absorption.
Water, electrolytes, glucose and other organic nutrients are reabsorbed in the convoluted tubule (proximal and distal sections) via passive and active transport. The epithelial membrane has carrier sites where active transport occurs. Active transport is where chemical energy is used to transport substances against concentration gradients – in this case, substance such as calcium, chloride, phosphate, potassium and sodium. Some ions (including chloride and sodium) can also be absorbed by passive as well as active transport mechanisms, depending on the location of the transport site within the nephron. There are some filtrate constituents (e.g. amino acids and glucose) that are generally completely reabsorbed and so don’t normally appear in urine unless blood levels are excessive (see below). On the other hand, nitrogenous waste products (e.g. creatinine, uric acid and urea) are are only reabsorbed in very limited amounts.
The renal threshold (or transport maximum) represents the maximum capacity that the kidneys have for reabsorption of a given substance. An example is glucose (mentioned above). If blood glucose level rises above the transport maximum (around 9 mmol/l or 160 mg/dL), glucose will appear in the urine (a common symptom of diabetes). The reason that this happens is because the carrier sites are all occupied and the active transport mechanism becomes overloaded. Hormones can also regulate the level of reabsorption. For instance, parathyroid hormone increases blood calcium levels and calcitonin (produced by the thyroid gland) lowers calcium levels.

There is a loop called the medullary loop (also called the loop on Henle) located at the bottom of the tubule and by this point around 30-40% of the filtrate has already been reabsorbed. The principal function of this loop appears to be the recovery of sodium chloride and water. As a so-called “counter-current multiplier”, the loop on Henle acts as system to transport material across a membrane which separates two countercurrent multiplier tubes (the proximal and distal sections of the convoluted tubule). It achieves this through its “hairpin” shape which means a concentration can be achieved in the fluid within the hairpin bend, relative to the inflow and outflow fluid concentrations, which is much greater than the transport mechanism could produce between the two sides of the membrane at any other point. At this stage, about another half of the remaining filtrate is reabsorbed (mainly water, chloride and sodium). By the time the filtrate reaches the distal convoluted tubule – “distal” means “outer” or “distant” – only around 15– 20% of the original filtrate remains. Naturally, the filtrate’s composition is by now significantly altered. At the distal convoluted tubule more electrolytes are reabsorbed (particularly sodium). From here, the filtrate will be quite dilute as it enters the collecting ducts, whose main function is to reabsorb water in accordance with bodily requirements.
Secretion describes this process where the filtrate absorbed in the glomerulus flows through the tubule as nutrients and water are reabsorbed into capillaries while, at the same time, waste ions and hydrogen ions pass from the capillaries into the tubule. The “secreted” ions combine with the remaining filtrate and become urine. After the above-mentioned stage when the urine flows out of the nephron tubule into the collecting duct, it then passes out of the kidney through the renal pelvis, into the ureter, and down into the bladder.
Thus, the kidney nephrons have processed blood to create urine through a process of filtration, reabsorption, and secretion. The resulting urine is around 95% water and 5% waste products (the nitrogenous wastes including urea, creatinine, ammonia, and uric acid, along with some ions of calcium, hydrogen, potassium and sodium).
How ADH controls water removal from the body.
In very hot, dry conditions (low humidity), a person can drink large amounts of fluid, but still excrete small quantities of hypertonic urine – why is this?
Antidiuretic hormone or ADH (secreted by the posterior pituitary) controls the amount of water removed from the body by increasing the permeability of both the distal convoluted tubules and the collecting tubules. The result of this is that there is increased water reabsorption back into the bloodstream from the glomerular filtrate. A negative feedback system controls ADH secretion.
The reason why only small quantities of hypertonic urine are excreted, even though a person drinks large amount of fluid during very hot, dry conditions (i.e. conditions of low humidity), is probably because the person is experiencing a degree of dehydration. This results in the body retaining as much water as possible and the likely production of hypertonic urine – that is, urine with a higher concentration of waste products (solute) to water (solvent) than normal. Part of the reason for this is that the person will be sweating a lot and this will be causing the body’s temperature and pH regulation systems to be protecting homeostatic levels. Drinking lots of water would normally, in ambient or cold and damp conditions, produce hypotonic urine (that is, a lower than normal concentration of waste products to water.
The individual processes involved in the retention of water (or, conversely, the production of hypertonic urine) involve the interaction of the loop of Henle and its nearby collecting tubule. Dehydration causes the kidneys to aim for maximal water reabsorption. The main driving force for water reabsorption is the urea concentration gradient created by the loop of Henle. Because of the impermeability of the thick segment to water molecules and the high reabsorption of solutes, the loop of Henle creates an osmotic gradient (from the cortex to the hypertonic medulla). Sodium (NA+), chlorine (Cl-) and urea have an important role in causing hypertonicity of the renal medulla to reach it maximum. At this point, production of ADH is increased (reabsorbing more water), and urea circulates in the renal medulla, thereby increasing its hypertonicity. Aquaporins are integral membrane proteins that are highly selective for water, and thus allow water to flow more easily across membranes – almost at diffusion rate. Aquaporins are relevant in this context because they allow water to flow from the collecting tubule to the nearby medulla. It is because the the close proximity of the loop of Henle and the collecting tubule that such a high efficiency for water reabsorption exists. The concentrating of urine (making it hypertonic) is not about leaving more ions in water, it is actually more about reabsorbing the maximal amount of water back into the bloodstream.
References
- What is Gout? The Arthritis Foundation. [↩]
- Ann Rheum Dis. Ann Rheum Dis. 2014 Feb 1; 73(2): 385–390. Published online 2013 Jan 23. doi: 10.1136/annrheumdis-2012-202589. Low-dose aspirin use and recurrent gout attacks. Yuqing Zhang, Tuhina Neogi, Clara Chen, Christine Chaisson, David J Hunter, and Hyon Choi. [↩]
- Obesity, weight change, hypertension, diuretic use, and risk of gout in men: the health professionals follow-up study. Choi HK, Atkinson K, Karlson EW, Curhan G. Arch Intern Med. 2005 Apr 11; 165(7):742-8. [↩] [↩]
- Drug.com. Thiazide diuretics. What are Thiazide diuretics? [↩]
- N Engl J Med. 1989 Aug 3;321(5):287-92. Cyclosporine-induced hyperuricemia and gout. Lin HY1, Rocher LL, McQuillan MA, Schmaltz S, Palella TD, Fox IH. [↩]
- Ann Intern Med. 2012 Aug 21;157(4):233-41. doi: 10.7326/0003-4819-157-4-201208210-00003. Low-level lead exposure and the prevalence of gout: an observational study. Krishnan E1, Lingala B, Bhalla V. [↩]
- Urea cycle: Wikipedia, the free encyclopedia [↩]
- MediCalc® Uric Acid Conversion Tool. [↩]
- convertunits.com: Mole Conversion Tool. [↩]
- Podcast: A Primary Care Primer with Dr Joseph Lieberman. [↩]
- CE Central: Joseph A. Lieberman III, MD, MPH [↩]
- Podcast: Gout: A Primary Care Primer. Summary. [↩]
- Gout epidemiology: results from the UK General Practice Research Database, 1990-1999. Mikuls TR, Farrar JT, Bilker WB, Fernandes S, Schumacher HR Jr, Saag KG. Ann Rheum Dis. 2005 Feb; 64(2):267-72. [↩]
- Epidemiology of gout in women: Fifty-two-year followup of a prospective cohort. Bhole V, de Vera M, Rahman MM, Krishnan E, Choi H. Arthritis Rheum. 2010 Apr; 62(4):1069-76. [↩]
- Fam AG. Gout, diet and the insulin resistance syndrome. J Rheumatol. 2002;29(7):1350–1355. [↩]
- Choi HK, Ford ES. Prevalence of the metabolic syndrome in individuals with hyperuricemia. Am J Med. 2007;120(5):442–447. [↩]
- Choi HK, Ford ES, Li C, et al. Prevalence of the metabolic syndrome in patients with gout: the Third National Health and Nutrition Examination Survey. Arthritis Rheum. 2007;57(1):109–115. [↩]
- Mayo Clinic: Do diuretics taken to control high blood pressure sometimes cause gout? Answers from Dr April Chang-Miller [↩]
- Intake of purine-rich foods, protein, and dairy products and relationship to serum levels of uric acid: the Third National Health and Nutrition Examination Survey. Choi HK, Liu S, Curhan G. Arthritis Rheum. 2005 Jan; 52(1):283-9. [↩] [↩]
- Arch Intern Med. 2009 Mar 9;169(5):502-7. doi: 10.1001/archinternmed.2008.606. Vitamin C intake and the risk of gout in men: a prospective study. Choi HK, Gao X, Curhan G. [↩]
- Arthritis Rheum. 2007 Jun;56(6):2049-55. Coffee consumption and risk of incident gout in men: a prospective study. Choi HK1, Willett W, Curhan G. [↩]
- Ann Rheum Dis. 2012 Sep; 71(9): 1448–1453. Purine-rich foods intake and recurrent gout attacks. Yuqing Zhang, Clara Chen, Hyon Choi, Christine Chaisson, David Hunter, Jingbo Niu, and Tuhina Neogi. [↩]
- British Society for Rheumatology and British Health Professionals in Rheumatology guideline for the management of gout. Jordan KM, Cameron JS, Snaith M, Zhang W, Doherty M, Seckl J, Hingorani A, Jaques R, Nuki G, British Society for Rheumatology and British Health Professionals in Rheumatology Standards, Guidelines and Audit Working Group (SGAWG). Rheumatology (Oxford). 2007 Aug; 46(8):1372-4. [↩]
- Arthitis-health: What Are Purines? [↩]
- Choi HK, Willett W, Curhan G. Fructose-rich beverages and risk of gout in women. JAMA. 2010;304(20):2270-8. [↩] [↩]
- Nutritional/Brewers Yeast & CD [↩]
- Arthritis-health: Gout Prevention Diet [↩]
- Mayo Clinic: Mayo Clinic. Nutrition and Healthy Eating: Gout diet. August 10, 2011. [↩]
- Schlesinger N. Dietary factors and hyperuricaemia. Curr Pharm Des. 2005;11(32):4133-8. Review. PubMed PMID: 16375734. [↩]
- Jacob RA, Spinozzi GM, Simon VA, Kelley DS, Prior RL, Hess-Pierce B, Kader AA. Consumption of cherries lowers plasma urate in healthy women. J Nutr. 2003 Jun;133(6):1826-9. PubMed PMID: 12771324. [↩]
- Gout Treatment with a Cherry on Top. Michael Greger M.D. FACLM June 11th, 2014 Volume 19 [↩]
- How to Treat Gout With Diet. Written By Michael Greger M.D. FACLM on October 8th, 2015 [↩]
- L M McCune, C Kubota, N R Stendell-Hollis, C A Thomson. Cherries and health: A review. Crit Rev Food Sci Nutr 2011 51(1):1 – 12. [↩]
- Y H He, J Zhou, Y S Wang, C Xiao, Y Tong, J C O Tang, A S C Chan, A P Lu. Anti-inflammatory and anti-oxidative effects of cherries on Freund’s adjuvant-induced arthritis in rats. Scand J Rheumatol. 2006 35(5):356 – 358. [↩]
- D S Kelley, R Rasooly, R A Jacob, A A Kader, B E Mackey. Consumption of Bing sweet cherries lowers circulating concentrations of inflammation markers in healthy men and women. J Nutr 2006 136(4):981 – 986. [↩]
- D S Kelley, Y Adkins, A Reddy, L R Woodhouse, B E Mackey, K L Erickson. Sweet bing cherries lower circulating concentrations of markers for chronic inflammatory diseases in healthy humans. J Nutr 2013 143(3):340 – 344. [↩]
- L W Blau. Cherry diet control for gout and arthritis. Tex Rep Biol Med. 1950;8(3):309-11. [↩]
- Y Zhang, T Neogi, C Chen, C Chaisson, D J Hunter, H K Choi. Cherry consumption and decreased risk of recurrent gout attacks. Arthritis Rheum 2012 64(12):4004 – 4011. [↩]
- Y Zhang, C Chen, H Choi, C Chaisson, D Hunter, J Niu, T Neogi. Purine-rich foods intake and recurrent gout attacks. Ann Rheum Dis 2012 71(9):1448 – 1453. [↩]
- Jacob RA, Spinozzi GM, Simon VA, Kelley DS, Prior RL, Hess-Pierce B, Kader AA. Consumption of cherries lowers plasma urate in healthy women. J Nutr. 2003 Jun;133(6):1826-9. PubMed PMID: 12771324. [↩]
- A C Gelber, D H Solomon. If life serves up a bowl of cherries, and gout attacks are the pits: Implications for therapy. Arthritis Rheum 2012 64(12):3827 – 3830. [↩]
- Nutritionfacts.org article by Lori: Gout. [↩]
- Alkaline Diet. Topic summary contributed by volunteer: Allen. Nutritionfacts.org article. [↩]
- R Siener, A Hesse. The effect of a vegetarian and different omnivorous diets on urinary risk factors for uric acid stone formation. Eur J Nutr. 2003 Dec;42(6):332-7. [↩]
- naturallifeenergy.com: An Alkaline Diet And Gout. BY AQIYL ANIYS | MARCH 3RD, 2016 | MODIFIED – JUNE 23RD, 2017. [↩]
- best-gout-remedies.com: Coriander for gout. [↩]
- C.U. Rajeshwari, S. Siri, B. Andallu Antioxidant and antiarthritic potential of coriander (Coriandrum sativum L.) leaves e-SPEN Journal 7(2012) e223-228. [↩]
- Cow’s Milk – But It Looks So Innocent. [↩]
- EFFECT OF CARBOHYDRATE AND FAT IN THE DIET ON URIC ACID EXCRETION. BY DAVID ADLERSBERG AND MAX ELLENBERG. From the Medical Service of Dr. George Baehr and the Laboratories of the Mount Sinai Hospital, New York. (Received for publication, February 16, 1939. [↩]
- Medications for Treating Gout. The Arthritis Foundation. [↩]
- IBD / Crohn’s Disease / Ulcerative Colitis & WFPB Diet Part 1 of 5 [↩]
- Healthline: Pseudogout definition. [↩]
- Arthritis Research UK: What are the Causes of Crystal Diseases? [↩]