Inflammatory bowel diseases (IBD’s), such as ulcerative colitis (UC) and Crohn’s disease (CD), are autoimmune conditions where your immune system attacks your own intestines. It’s thought that there’s no cure for such diseases, so all you can do is try to stay in remission for as long as possible between attacks. But can a plant-based diet help to both prevent and treat these debilitating conditions? This is a complex topic, so it has been covered in three parts. This is part 2 of 5.
(See Further Notes, below, for a more detailed explanation of IBD, UC & CD)
This article will look at some of the research related to plant-based diets as a whole, and then analyse in more detail those individual elements constituting a human diet in relation to IBD, UC & DC. These will include:
- Carbohydrates
- Protein
- Fat,
- Omega-6 PUFA (Polyunsaturated Fatty Acids)
- Omega-3
- Total PUFA intake
- Trans-fats
- Dairy
- MAP bacteria in Dairy
- Fibre
- Microflora (microbiota)
- Nutritional yeast
- Citrus Fruits
- Arachidonic acid
- Zinc
- IgG4
- Lipase
- Peppermint oil
- Turmeric (curcumin)
- Vitamin deficiencies – B9 (folate) & B12
Blog Contents
Plant-Based Diets and IBD (UC & CD)
Cutting out all animal foods and just eating healthy fruits and veg was shown to be highly effective in a study1 mentioned in an article by Dr Michael Greger, where he says: “maybe instead of doing all the fancy blood tests, we should just tell our patients to cut out meat and eggs and see how they do*, which would be consistent with both population studies2 that associate animal fat with an increased risk of inflammatory bowel diseases, as well as interventional studies3 4 5 showing that a plant-based diet, in which you cut meat down to like one serving every two weeks, can drop relapse rates as low as an extraordinary 8% over two years.”6
*All bold in quotes or elsewhere are my personal additions.
As stated elsewhere, the cause of CD and other diseases of the digestive system are not currently fully understood, but there are a number of theories about their aetiology (cause). One that’s gaining popularity is the emerging theory that CD is an abnormal immune response to the micro flora (bacteria) normally found in the gut. It’s noticeable that CD increased during the 20th century and shows no sign of abating7 as compared to the recent past. This suggests that there may be a link with dietary factors (increased consumption of animal protein/processed food) as well as allergens, smoking, or children not being exposed to various bacteria or viruses as they were in the past and thus building an immunity.
A 2015 study8 concluded that, whilst the precise mechanisms involved are complex and still require further research, epidemiology provides convincing evidence that a plant-based diet can provide therapeutic and/or preventive effects against pretty much all the current major chronic diseases, including IBD.9 10 3
Dr Renae Thomas (a moderator on nutritionfacts.org) recommends the following plant foods for CD: sweet potatoes, well cooked vegetables such as summer squash, well ripened or cooked fruit, rice and gluten free grains.11
Carbohydrates and IBD (UC & CD)
Research12 suggests that while refined and processed carbohydrates and intake of sweetened beverages are risk factors for IBD, complex carbohydrates including fruit, vegetables and fibre should be included in the diet to manage IBD. Soluble fibre from fruit may have a protective effect on CD13 High vegetable intake may be associated with decreased risk of UC 12 . The European Investigation into Cancer and Nutrition study reported that an increased consumption of sugar and soft drinks with low vegetable intake was positively associated with UC risk14 . Increased consumption of sweets is positively associated with CD and UC. 15
Dr John McDougall recommends16 that “Patients who are very ill may need to be put on very simple diets, supplemented with some drugs until the bowel quiets down. As soon as the patient can eat regular foods comfortably, he should be started on a starch-centered diet without wheat or citrus fruits initially (later these two kinds of plant foods may be tried). The elimination diet is the next approach for stubborn cases.”
Protein and IBD (UC & CD)
A large prospective cohort study completed over a 10.5-year period found that high protein intake, specifically animal protein (meat, not dairy products) was positively associated with an increased risk of IBD17 . A systematic review reported an association with high total protein intake with the development of UC and CD12 . High protein intake was associated with a 3.3-fold increased risk of IBD, suggesting a diet high in animal protein is a major risk factor for the development of IBD.
Fat and IBD (UC & CD)
Research in 197418 showed that removing dietary fat from the diet had significant benefits for the treatment of diseases of the small intestine. This finding was supported by the following research in 1997 and 2000:
- “Our findings suggest that dietary factors are important in the development of IBD. These factors may have a primary effect in predisposed subjects, or may modulate the effect of other as yet unidentified environmental factors. If and when our findings are confirmed by other investigators, they may be considered for practical application. For instance, in a family with one or more first degree relatives with IBD, a “prudent diet” may be recommended for uninvolved members of the family, particularly the children. This may, in the future, apply mainly to those family members shown to be genetically predisposed to IBD.”19
- “High intakes of mono- and polyunsaturated fat and vitamin B6 may enhance the risk of developing UC. Whether this observation is a true risk factor in the development of UC or rather a reflection of a certain dietary lifestyle needs to be investigated.”20
This short video21 by Dr Neal Barnard covers diet in relation to the following (often interrelated) topics:
- Gastroesophageal reflux disease
- Irritable bowel syndrome
- Lactose intolerance
- Crohn’s disease
- Gall stones
- Colon cancer
- Constipation
Amongst other useful and interesting nuggets of advice, Dr Barnard advises CD (as well as other IBD and IBS) sufferers to avoid:
- Dairy
- Artificial sweeteners
- Wheat
- Eggs
- Fatty foods
The reason that it’s important to avoid fat is that when fat touches the intestinal wall it causes the muscles to contract – a bit like squeezing both ends of a toothpaste tube at the same time – ouch!).
Omega-6 and IBD (CD & UC)
Dietary n-6 PUFA (polyunsaturated fatty acid), in particular linoleic acid, have been implicated in the aetiology of IBD. Dietary n-6 PUFA’s are essential fatty acids present in high amounts in red meat, cooking oils (safflower and corn oil) and margarines.
A 4-year long prospective cohort study in 200922 concluded that: “The data support a role for dietary linoleic acid in the aetiology of ulcerative colitis. An estimated 30% of cases could be attributed to having dietary intakes higher than the lowest quartile of linoleic acid intake.” Foods high in omega-6 include: vegetable oils, salad dressings, crisps and chips, fried fast food, cookies, pastries, crackers, pork, sausages, bacon, chicken, dairy, eggs, nuts. The researchers explain the reason why omega-6 is harmful: “Dietary linoleic acid, an n-6 polyunsaturated fatty acid, is metabolised to arachidonic acid, a component of colonocyte membranes. Metabolites of arachidonic acid have pro-inflammatory properties and are increased in the mucosa of patients with ulcerative colitis.”
In his book Fast Food Genocide23, Dr Joel Fuhrman talks about the effects of some of the usual fried foods: “Think of French fries or doughnuts, for example, which are just high-glycemic carbohydrates fried in [omega 6] oil. That’s suicide on a plate. Such foods can generate inflammation, accelerate aging, create serious autoimmune diseases such as Crohn’s disease and ulcerative colitis, and have dramatic damaging effects on brain function.”
He references a 2002 study24 into the effects of potato glycoalkaloids (compounds that add a bitter flavour) when they are fried. The researchers state: “Disruption of epithelial barrier integrity is important in the initiation and cause of inflammatory bowel disease (IBD). Glycoalkaloids, solanine (S), and chaconine (C) are naturally present in potatoes, can permeabilize cholesterol-containing membranes, and lead to disruption of epithelial barrier integrity. Frying potatoes concentrates glycoalkaloids. Interestingly, the prevalence of IBD is highest in countries where fried potatoes consumption is highest.”
Omega-3 and IBD (UC & CD)
It has been speculated that the intake of long-chain n-3 PUFA‘s (DPA – docosapentaenoic acid, EPA – eicosapentaenoic acid, DHA – docosahexaenoic acid), known as omega-3’s, may be of benefit to patients with IBD. The beneficial effects are believe to be derived from the anti-inflammatory properties of n-3 PUFA’s.
Many people assume that fish oils, being rich in these these PUFA’s, are therefore a wise dietary choice. However, clinical and experimental studies have shown conflicting results. In 2008, a study25 reviewed two randomised, double-blind, placebo-controlled studies (Epanova Program in Crohn’s Study 1 [EPIC-1] and EPIC-2) conducted between January 2003 and February 2007 at 98 centres in Canada, Europe, Israel, and the United States. The researchers concluded that: “In these trials, treatment with omega-3 free fatty acids was not effective for the prevention of relapse in Crohn disease.”
Other meta-analyses have also failed to show benefit with supplementation with fish oils in the maintenance of remission in CD and UC:
- A 2011 review26 concluded that: “There are insufficient data to recommend the use of omega 3 fatty acids for maintenance of remission in CD and UC.“
- A 2012 review27 concluded that “Overall, available data do not allow supporting the use of omega-3 fatty acid supplementation for the treatment of both active and inactive IBD. Negative results are quite consistent in trials assessing the use of omega-3 fatty acids to maintain disease remission, particularly UC, and to a lesser extent CD.”28
- A 2014 review29 concluded that: “Evidence from two large high quality studies suggests that omega 3 fatty acids are probably ineffective for maintenance of remission in CD. Omega 3 fatty acids appear to be safe although they may cause diarrhea and upper gastrointestinal tract symptoms.“
Dietary intake of n-3 PUFAs were inversely associated with risk of UC, whereas no association has been found with CD30.
Total PUFA Intake and IBD (CD)
The total ratio of n-3 PUFA: n-6 PUFA found in the diet is thought to be important, specifically, a higher amount of n-3 in relation to n-6 than is normal in the SAD (Standard american Diet). This was supported by three studies:
- A 2007 case-control study31 which considered that a high n-3 : n-6 ratio in the diet is inversely associated with the risk of IBD; concluding that: “Our findings indicate that an imbalance in consumption of fatty acids, vegetables, and fruits is associated with increased risks for CD among Canadian children.“.
- A 2010 prospective cohort study32 concluded in relation to UC: “Total dietary n-3 PUFAs, EPA, and DHA, particularly DHA were associated with protection from UC in a cohort aged over 45 years. If the association is causal, then increasing the population’s intake of n-3 PUFAs from oily fish may help prevent UC.” There are serious problems with n-3 intake from fish and these are covered to some extent in another blog.33
- A 2010 dietary intervention trial34 concluded: “N-3DP [“n-3 PUFA food exchange table”] significantly increased the erythrocyte [a cell that contains haemoglobin and can carry oxygen to the body] membrane n-3/n-6 ratio in IBD patients, and this ratio was significantly higher in the remission group, suggesting that n-3DP alters the fatty acid composition of the cell membrane and influences clinical activity in IBD patients.“
Further analysis of the above European Investigation into Cancer and Nutrition study over five years35 found: “…a possible increased risk with a higher total polyunsaturated fatty acid intake. A biological mechanism exists in that polyunsaturated fatty acids are metabolised to pro-inflammatory mediators.”
The latter research as also supported by a systematic review of the relevant literature that examined pre-illness intake of nutrients and subsequent development of UC.12 Researchers’ concluded that: “High dietary intakes of total fats, PUFAs, omega-6 fatty acids, and meat were associated with an increased risk of CD and UC. High fiber and fruit intakes were associated with decreased CD risk, and high vegetable intake was associated with decreased UC risk.”
A 2005 case-control retrospective study in CD15 , already referenced above, found that increased total PUFA consumption was positively associated with CD risk. Researchers concluded: “...the intake of total fat, monounsaturated fatty acids and polyunsaturated fatty acids, vitamin E [supplements], and n-3 [omega-3 PUFA] and n-6 fatty acids was positively associated with CD risk… the present findings suggest the importance of dietary factors for IBD prevention.”
Trans Fats and IBD (UC & CD)
The Nurses’ Health Study cohorts reported high, long-term intake of trans-unsaturated fatty acids (commonly abbreviated to ‘trans-fats’) was associated with a trend towards an increased incidence of UC but not CD36 The researchers concluded: “A high intake of dietary long-chain n-3 PUFAs may be associated with a reduced risk of UC. In contrast, high intake of trans-unsaturated fats may be associated with an increased risk of UC.”
A 2016 study37 stated that: “the most striking finding was an increased relative risk of both Crohn’s disease and ulcerative colitis associated with consumption of fast foods: the relative risk associated with consumption of fast foods at least two times a week was estimated at 3.4 (95% confidence interval = 1.3-9.3) for Crohn’s disease and 3.9 (95% confidence interval = 1.4-10.6) for ulcerative colitis. Although coffee seemed to provide a protective effect for both diseases…”
An increased relative risk of developing IBD has also been associated with frequent intake of fast foods (fast foods are high in trans-unsaturated fatty acids). A 2016 Australian population-based study38 found that: “The factors associated with CD included smoking…childhood events, including tonsillectomy…chicken pox infection…and pre-diagnosis intake of frequent fast food. In UC, the risk factors included smoking…and pre-diagnosis intake of frequent fast food [interestingly], and high caffeine intake was protective. Other protective exposures for UC included high fruit intake… and [again, interestingly] having pets as a child.”
Dairy and IBD (UC & CD)
There is considerable controversy in the area of dairy and IBD. I will simply present a few of the opinions here.
The European Investigation into Cancer and Nutrition study found that individuals that consumed milk had significantly reduced odds of developing CD, suggesting a protective effect with dairy product consumption39 N.B. The conclusions from this study were: “Milk consumption may be associated with a decreased risk of developing CD, although a clear dose-response relationship was not established. Further studies are warranted to confirm this possible protective effect.” This is supported by a case-control study in children that demonstrated that consumption of dairy products was not associated with CD31 .
A commentator on the nutritionfacts.org website, Dr. Lester Castro Friedlander, explained about a condition in dairy cows called Johnnes Disease: “[the] USDA allows these diseased cows to go to slaughter for human consumption. In various research thru out the years, it has been suggested that there might be a link between Johnnes Disease [40 ] in dairy cows to Crohn’s Disease in Humans. According to research done in Great Britan (sic.), The milk of dairy cows with Johnnes Disease was pasteurized for Human Consumption. After pasteurization, the bacteria that causes Johnnes Disease was still present. Pasteurization did not kill the bacteria.”41
Research as long ago as 196142 suggested that milk provokes UC.
Just after this, in 196543 , further research confirmed these findings and concluded that “…a milk-free diet is beneficial to about one in five patients with ulcerative colitis, with a suggestion that the proportion may be higher in patients in their first attack of the disease.”
Dr Greger considers that “Just cutting out milk can sometimes help with inflammatory bowel disease.”44
MAP in Dairy and IBD (UC & CD)
Following up on Dr Friedlander, it is clear that there is evidence that CD is potentially caused by a bacteria in dairy called Mycobacterium Avium Paratuberculosis (MAP)45 that seems to colonise the bowel and is difficult to eradicate even with pasteurisation.46 The author of this study concludes: “It is likely that MAP is the cause of a range of human gastrointestinal diseases including irritable bowel syndrome47 48 as well as ulcerative colitis and Crohn’s disease. If we accept that both ulcerative colitis and Crohn’s disease are caused by a specific infectious microorganism, MAP, a long overdue transformation will take place in the prevention and treatment of these diseases.”
Further research in 201549 found that: “Increasing research evidence links human inflammatory bowel disease to MAP infection in food animals. Crohn’s disease may well be a by-product of modern animal husbandry practices in use by the dairy industry.”
Fibre and IBD (UC & CD)
Contrary to the long-held – and erroneous – belief, fibre has been shown to be beneficial for CD.8
Dr John McDougall says16 “Bowel contents must be changed continually in order to get long lasting significant improvement. The simple increase of the fiber content in the foods eaten has been shown to reduce the frequency of attacks and to improve symptoms in many patients. A starch-based diet, which is inherently high in fiber, is highly effective at alleviating the distress from this condition. One step further in effective therapy is to avoid the foods that most often cause allergies–dairy products, eggs, chocolate, wheat, and citrus fruits, as well as fats of all kinds. If this approach is not successful, the final step toward diagnosis is to use an elimination diet.”
In 197950 , researchers claimed that “Treatment with a fibre-rich, unrefined-carbohydrate diet appears to have a favourable effect on the course of Crohn’s disease and does not lead to intestinal obstruction.”
A 2016 population-based case-control study37 of IBD and dietary habits, mentioned above, found that: “The relative risk of Crohn’s disease was increased for subjects who had a high (55 gm or more per day) intake of sucrose…and was decreased for subjects who had a high (15 gm or more per day) intake of fiber…”
Dr Neal Barnard states21 that high fibre foods were once said to be bad for you, but now they are considered to be excellent for CD. He recommends well-cooked beans, vegetables, fruits, and whole grains, while possibly going easy on raw veg and fruits until one is sure that they do not cause unpleasant symptoms.
Microflora and IBD (UC & CD)
The food you eat determines not only the health of the trillions of bacteria living in your gut, but also the types of bacteria that live there. If you eat an animal based diet, full of processed “Frankenfoods”51 and devoid of fibre, your gut bacteria are not going to protect you as well as if you eat a whole food plant-based diet with no added salt, oil or sugar.52 The result of this can be the breakdown in the overall health of your gastrointestinal (GI) tract.53 54 As the researchers state in the previously-referenced study: “there is a need for dedicated dietary intervention studies in humans to differentiate between correlation and causality.”
Research is still in its infancy, however there is growing evidence for an association between IBD and an alteration in the gut microbiota. The Westernised diet (often called the SAD – Standard American Diet), characterised by increased consumption of PUFA, animal protein, and sugar as well as decreased consumption of fibre has been implicated as factor contributing to dysbiosis (also called dysbacteriosis – is a term for a microbial imbalance or maladaptation on or inside the body, such as an impaired microbiota). 55 56 57
Diets that produce a low diversity of the microbiota has been linked to a variety of chronic diseases 58
In the following section of a video59 about leaky gut syndrome (LGS) by Dr Michael Klaper, he advises against antibiotics, since they kill ALL bacteria – the good and the bad. He also talks about the difference between eating sugar as a flavouring versus eating sugar as a food, explains what happens when you kill off “good” bacteria in your body, and how to defend yourself against “modern life’s assault on friendly intestinal bacteria.” He advises the omission all dairy and probably all wheat (certainly highly refined versions), as well as all other animal products.
Dr Klaper says that the intestinal barrier function (vital to intestinal health and always compromised with IBD) can be repaired through taking three supplements:
1. The flavanoid quercetin (500-1000 mg twice daily for 6 weeks). Longer-term, quercetin is best consumed in quercetin-rich foods: apples, peppers, dark cherries and berries (blueberries, bilberries, blackberries and others), tomatoes, cruciferous vegetables (including broccoli, cabbage and sprouts), leafy green vegetables (including spinach and kale), and citrus fruits. Of course, quercetin is also found in red wine – but he STRONGLY advocates that ZERO alcohol is ever consumed in order to preserve the good gut bacteria. So, non-alcoholic red wine is an option.
2. The amino acid glutamine, taken initially as a supplement (500-1000 mg twice daily for 6 weeks). Long-term, glutamine is found in all foods, but these are good sources: cabbage, spinach, kale, parsley, beets and beans.
3. Probiotic supplements (such as lactobacillus acidophilus). In this respect, Dr Greger advises that our food provides the probiotics we need60. This makes a lot of sense when the role of butyrate-producing bacteria52 from dietary fibre is taken into account.
Further details on Dr Klaper’s video are available.61
The following video ‘Ulcerative Colitis, Crohn’s Disease and Leaky Gut Syndrome with Michael Klaper, M.D.’62 covers much of the same material as above, although with more graphics and additional detail.
Nutritional/Brewer’s Yeast & CD
Bakers have both the highest CD incidence rates and CD mortality rates.
Yeast (and possibly citrus – see below) may produce some of the biggest immune responses in CD patients (even more than milk). This has been known for a long time. A 1992 study63 draw the understated concluded that: “…dietary yeast may affect the activity of Crohn’s disease.”
The implications of this and other research studies were further discussed in a Dr Greger video44 : “19 patients with Crohn’s disease ate their regular diet, and then switched to a yeast-exclusion diet. And, there was a significantly higher CDAI (Crohn’s disease activity index), assessing symptoms like abdominal pain and diarrhea during the period when they were eating yeast, compared to the yeast-free period. So, worse disease activity with yeast than without…After placing them on the new yeast-free diet, they then challenged them with a placebo or a capsule of yeast. A tiny amount of yeast, like giving them a quarter-teaspoon of nutritional yeast a day, and that made them worse—suggesting “yeast may [indeed] be important in the [disease process] of Crohn’s disease.” Now, for the vast majority of people, yeast is not a problem, but “in susceptible individuals [it may trigger] an abnormal immune response” in the gut. But wait, I thought it was the paratuberculosis bug [see MAP above] that was considered a trigger for Crohn’s disease. Well, hey, maybe infection with paraTB is what “induces [the] hypersensitivity response to dietary yeast.” Who knows? The bottom line is that people with Crohn’s disease should not go out of their way to add baker’s, brewer’s, or nutritional yeast to their diet.”
The following Dr Greger video64 Does Nutritional Yeast Trigger Crohn’s Disease? is worth watching if you require further details. N.B. The references he cites are listed below the video.
Citrus Fruits
A 2002 study65 at St Mark’s Hospital, Middlesex, England looked at the responses of CD patients to skin testing and rectal exposure to six food
antigens (cereal, cabbage, citrus, milk, yeast and peanut). They found that: “…patients with Crohn’s disease have a markedly increased response to food antigens, demonstrated both in vivo and in vitro, compared with healthy subjects. Markedly increased responses were observed to all the food antigens tested in the Crohn’s disease patients , although the particularly stringent statistical criteria which were applied suggested that the most significant responses occurred to yeast and citrus fruits.”
It is worth noting, however, that citrus fruits are known to be anti-inflammatory and are an important part of a balanced diet if tolerated by the individual66. This is also pointed out by Dr Michael Greger.67 68
Arachidonic Acid and IBD
Dr Greger says in his video Inflammatory Remarks about Arachidonic Acid69 that: “Inflammatory bowel disease risk has been tied to arachidonic acid, which may partially explain the animal protein connection given the levels in chicken and eggs.” He talks further about inflammation in his article Treating Crohn’s Disease With Diet70
The following table shows what the NIH (US National Institutes of Health) found were the highest sources of arachidonic acid in the US diet between 2005 and 2006:71
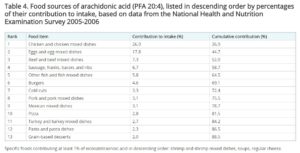
Zinc and CD
Research72 into the link between low zinc absorption and CD looked at two large prospective cohorts of women and found that intake of zinc was inversely associated with risk of CD but not UC. This association was particularly strong for dietary zinc and persisted with long-term zinc intake. Considerable mechanistic plausibility supports this association, including the effect of zinc on autophagy (the consumption of the body’s own tissue as a metabolic process occurring in starvation and certain diseases) and microbial clearance, innate immune response and maintenance of the integrity of the epithelial barrier73 (the sensitive cell lining of the intestines).74 75 76 77 78 79 80 81 82
Low plasma zinc has been found in patients with alcoholic cirrhosis, CD and coeliac disease.83 The signs of zinc deficiency include a rash on the face, groin, hands and feet and diarrhoea. These symptoms can easily be reversed by eating zinc-rich foods.
Plant foods high in zinc include:
- Beans and legumes
- Nuts and seeds (the best being pumpkin seeds, sunflower seeds, walnuts, cashews, almonds, pecans, chia seeds, and hemp seeds)
- Oats
- Wheat germ (careful if you have a wheat sensitivity)
- Nutritional yeast (to be avoided for IBD sufferers, particularly CD)
- Tofu
- Spinach
- Mushrooms
- Asparagus
- Broccoli
IgG4 & DC
IgG4 is an immunoglobulin (that is, a class of protein present in the serum and cells of the immune system, which functions as an antibody) that is seen to be stimulated in CD84 as well as in those with wheat allergies.85 There have been some successes in excluding certain foods from the CD sufferer’s diet. This research86 removed sixteen different foods (milk87 88 89 , peanuts, soya, shrimp, egg90 91 , tomato, pork, beef, cod, fish, potato, wheat, yeast, cheddar cheese, lamb and rice).
Results indicated that removing milk, pork, beef and egg was most strongly associated with symptom improvement.
Research in 201792 found that: “Patients with IBS had significantly higher IgG4 titers [the concentration of an antibody, as determined by finding the highest dilution at which it is still able to cause “clumping” or agglutination of the antigen] to wheat, leek and taro compared to those of controls. Serum IgG4 titers to ginger, cocoa, walnut, white radish, onion, and lettuce in IBS patients tended to be higher than controls. IgG4 titers to wheat, gluten and gliadin [a class of protein present in wheat and several other cereals within the grass genus Triticum] in the diarrhea subgroup, and lettuce, leek and taro in the non-diarrhea subgroup tended to be higher compared with controls. The number of subjects with positivity to apple, orange, lettuce, and leek was significantly higher in IBS patients than controls. The number of subjects with positivity to apple, orange, gluten, and gliadin in the diarrhea subgroup, and egg white, pineapple, soybean, lettuce, and leek in the non-diarrhea subgroup was significantly higher compared with controls.”
Lipase & CD
Lipase is primarily produced in the pancreas but it is also produced in the mouth and stomach. The majority of people produce enough pancreatic lipase, but people with cystic fibrosis, CD and coeliac disease may not have enough lipase to get the nutrition they need from food.93 Pancreatitis may occur as a result of CD.94
Peppermint Oil & IBS
In the Dr Greger video Peppermint Oil for IBS (Irritable Bowel Syndrome)95 he says: “Nine randomized placebo-controlled studies [These nine studies and other related studies are listed underneath the video] have found peppermint oil to be a safe and effective treatment for irritable bowel syndrome. A few adverse events were reported, but were mild and transient in nature, such as a peppermint taste, peppermint smell, and a cooling sensation around one’s bottom; whereas in some of the head-to-head peppermint versus drug studies, some of the drug side effects were so unbearable they had to drop out of the study. This suggests that it might be a reasonable approach for clinicians to treat IBS patients with peppermint oil as a first-line therapy, before trying anything else.”
Whilst IBD shouldn’t be confused with IBS (since IBS is a much less serious affliction than either CD or UC and doesn’t involve inflammation or appear to have a physiological basis), is there a use of peppermint oil in cases of IBD?
This 2017 study96 stated that: “No definite conclusions can be drawn as to the efficacy of…herbal medicinal products [including peppermint oil] in IBD patients due to the low number of studies and the lack of randomized controlled trials that replicate the results obtained in the individual studies conducted so far.”
So the jury is out on its efficacy in treating IBD (CD & UC) until here are further studies.
Turmeric (curcumin) and UC
Curcumin is the active ingredient found in turmeric, a common Indian spice. The yellow pigment (curcumin) present in turmeric has been shown to exhibit numerous anti-inflammatory properties97 . Curcumin is mainly administered orally in the form of capsules filled with its powder since some people find it’s taste rather bitter when added to food – although this is my personal preferred method.
A 2006 randomised controlled trial98 with UC patients showed significantly reduced relapse rates.
A systematic review99 and a Cochrane review100 concluded that: “Curcumin may be a safe and effective therapy for maintenance of remission in quiescent [a state or period of inactivity or dormancy] UC when given as adjunctive therapy along with mesalamine or sulfasalazine.”
In any event, it would appear that turmeric is a sensible addition to try with IBD in order to see if there is a negative reaction to i. If not, then there are many benefits from a daily dose (just a quarter of a teaspoon does it!) – as can be seen clearly if you refer to an article on Turmeric101 by Dr Greger, where he also lists several informative videos extolling the anti-inflammatory, anti-cancer and other benefits of turmeric.
Vitamin deficiencies – B9 (folate) & B12 and IBD (UC & CD)
A 2017 study102 concluded that: “…vitamin B12 and folate deficiencies were more common in patients with CD than in UC patients and controls. Prior ileal [the third and final part of the small intestine] or ileocolonic [the location of the valve where the ileum opens into the large intestine] resection was a risk factor of serum vitamin B12 abnormalities, and a disease duration within 5 years was a risk factor of low serum folate levels in CD patients.”
A 2013 study103 concluded that: “A significant proportion of patients with Crohn’s disease suffer from vitamin B12 and/or folate deficiency, suggesting that regular screening should be performed, with closer monitoring in patients with ileal resection or active disease.”
Of course, B12 is a vital supplement for those eating a WFPB diet, although it has been shown that most dietary groups in (including omnivores) in the western world show widespread B12 deficiency.104 The writer stated that: “Nearly two-fifths [40%] of the U.S. population may be flirting with marginal vitamin B12 status…” And this was back nearly two decades ago.
Self-imposed dietary restrictions may have an important impact on nutritional status. For example, 40–78% of IBD patients are believed to have inadequate folate intake. 105 Folate is found predominately in leafy and non-leafy vegetables such as spinach, asparagus, and Brussels sprouts. 106 Patients avoiding these foods for fear of worsening symptoms may be vulnerable to inadequacies in their diet.
Closing Remarks
I hope the foregoing has shone some light on the dietary factors that may affect IBD (UC & DC). More research is being published each week on this topic and it will be regularly added to this article as it appears.
In Part 3 of 5
We will look at the historical and geographical occurrence of IBS (UC & DC), as well as some further methods (such as elimination diets, fasting etc) used for dealing with this group of debilitating diseases.
Further Notes107 on Irritable Bowel Disease (IBD), Crohn’s Disease (CD) & Ulcerative Colitis (UC)
IBD refers to diseases of a chronic or remitting/relapsing intestinal inflammation. This guideline describes UC and CD as the major forms of inflammatory bowel diseases of unknown cause (aetiology). Both diseases develop complicated pathology, again with unknown causes, and mainly affect the gastrointestinal tract, resulting in various clinical symptoms.
UC and CD are collectively referred to as IBD because the two diseases share common or similar features; however, disease location, morphology (form and structure), and pathophysiology (physiological changes associated with a disease) are clearly different between them, and they are considered to be independent diseases. Moreover, it is necessary to classify them because diagnostic procedures, therapeutic interventions, and follow-up observation are somewhat different. Notably, it is called ‘IBD unclassified’ when colonic lesions have the features of IBD which cannot be classified as UC or CD.
Patients with IBD often experience impaired daily quality of life (QOL) since both diseases can appear at young ages and progress into repeated cycles of chronic relapse and remission throughout life.
UC is a diffuse non-specific inflammatory disease of unknown cause that continuously affects the colonic mucosa proximal from the rectum and often forms erosions and/or ulcers. It frequently repeats cycles of relapse and remission during its course and may be accompanied by extraintestinal complications. When it extensively affects the large intestine for a long period of time, a risk of developing cancer increases [a].
CD is a chronic inflammatory disease of unknown cause, although there are theories: one theory suggests an abnormal immune response to gut bacteria; another suggests processed foods or recently emerged allergens may be responsible; additionally, there appears to be a genetic factor (gene NOD2) running in families.108 Other causal factors may include certain viral or bacterial infections, ethnicity, smoking and even the use of a treatment for acne scars (isotretinoin – although there are strong objections109 to this drug being responsible).
CD is characterised by discontinuously affected areas with transmural granulomatous inflammation (where intestinal walls are breached, resulting in an accumulation of immune cells called histiocytes – a type of tissue macrophage) and/or fistula (an abnormal connection between an organ and another structure). This is often referred to as a ‘leaky gut’, and as intestinal contents spill into the blood, and get attacked by white blood cells, the result may be all sorts of additional and serious health problems.
It can affect any region in the digestive tract from the mouth to the anus, but is more likely to involve the small intestine (the ileum) and large intestines (the colon and especially the ileocaecal valve) and the area around the anus (perianal region) [b]. The inflamed tissues become thick and swollen, and the inner surfaces of the digestive system may develop open sores (ulcers). Generally speaking it’s found in the colon some 30% of the time and 30% in the ileum/small intestine. Both sites have been shown, when taken together, to have an occurrence rate of approximately 40%.
CD is less common than UC. Both men and women are equally affected. CD can occur at any age but there may be peaks at 15 to 30 years old and between 50 and 70 years old. Even very young children can develop the disease. In fact, 20 per cent of CD patients are diagnosed before the age of 20. It has also been established that CD is more common among smokers. There are many more potential symptoms in CD than in many gastrointestinal diseases. Signs and symptoms tend to flare up multiple times throughout life.
The most common features of CD this condition are:
- Persistent diarrhoea
- Abdominal pain and cramping
- Loss of appetite
- Weight loss
- Anal problems
- Anaemia
- Fever
Some people with CD have blood in the stool from inflamed tissues in the intestine; over time, chronic bleeding can lead to a low number of red blood cells (anaemia). The inflammation in CD extends through the bowel wall leading to abscess formation or scarring and narrowing of the bowel known as stricture formation. In addition, it may develop extraintestinal complications in systemic organs such as the liver, joints, the skin, and the eyes. This makes CD a ‘multi-system’ or generalised disease.
In addition, symptoms will depend on the severity of the disease and which part of the bowel is affected.
Traditional nutritional opinion has been that there is no specific diet recommended for all patients with CD. Nutritionists and medical professionals have historically considered that general nutrition should concentrate on providing adequate macro- and micro-nutrients in an easily-absorbed form, especially after intestinal surgery. The object of management of the disease to date has been to treat periods of relapse in order to produce remission. The task then has been to maintain the disease in an inactive state. If complications develop they would then need to be treated specifically. The treatment of CD has always complex, difficult and has required specialist medical advice. Treatment has usually focused on a combination of dietary intervention and drugs. Because of the relative ineffectiveness of this approach, surgery has been required for the majority of patients.
The disease is named after Dr Burrill B Crohn, a New York doctor, who reported cases in the 1930’s.
The incidence of colorectal cancer (CRC) is significantly increased in UC patients who have extensive lesions for a long period of time, and it is also known that the incidence of cancers in the small and large intestines, especially in the rectum and anal canal region, is high in CD patients. Therefore, an efficient surveillance strategy for cancer development is normally expected to be established by the patient’s doctor. IBD is considered to be a disease that does not significantly affects the patients’ life prognosis, although IBD patients have slightly shorter life prognosis compared to normal individuals.
This is a must-watch video ‘Inflammatory Bowel Disease, Crohns, and Colitis with Pamela A. Popper‘.
References
- Lewis JD, Abreu MT. Diet as a Trigger or Therapy for Inflammatory Bowel Diseases. Gastroenterology. 2017 Feb;152(2):398-414.e6 [↩]
- Perm J. 2014 Fall; 18(4): 94. doi: 10.7812/TPP/14-117. Plant-Based Diets in Crohn’s Disease. Mitsuro Chiba, MD, Hideo Ohno, MD, Hajime Ishii, MD, and Masafumi Komatsu, MD [↩]
- Tuso PJ, Ismail MH, Ha BP, Bartolotto C. Nutritional update for physicians: plant-based diets. Perm J. 2013 Spring;17(2):61–6. DOI: http://dx.doi.org/10.7812/TPP/12-085 [↩] [↩]
- Takagi S, Utsunomiya K, Kuriyama S, et al. Effectiveness of an ‘half elemental diet’ as maintenance therapy for Crohn’s disease: a randomized-controlled trial. Aliment Pharmacol Ther. 2006 Nov 1;24(9):1333–40. [↩]
- Hirakawa H, Fukuda Y, Tanida N, Hosomi M, Shimoyama T. Home elemental enteral hyperalimentation (HEEH) for the maintenance of remission in patients with Crohn’s disease. Gastroenterol Jpn. 1993 Jun;28(3):379–84. [↩]
- Chiba M, Abe T, Tsuda H, Sugawara T, Tsuda S, Tozawa H, Fujiwara K, Imai H. Lifestyle-related disease in Crohn’s disease: Relapse prevention by a semi-vegetarian diet. World J Gastroenterol. 2010 May 28; 16(20):2484-95. [↩]
- Review: The role of diet in triggering human inflammatory disorders in the modern age. Huang EY, Devkota S, Moscoso D, Chang EB, Leone VA. Microbes Infect. 2013 Nov; 15(12):765-74. [↩]
- Perm J. 2015 Winter; 19(1): 58–61. doi: 10.7812/TPP/14-124. High Amount of Dietary Fiber Not Harmful But Favorable for Crohn Disease. Mitsuro Chiba. Tsuyotoshi Tsuji. Kunio Nakane. Masafumi Komatsu. [↩] [↩]
- Vegetarian diets, low-meat diets and health: a review. McEvoy CT, Temple N, Woodside JV. Public Health Nutr. 2012 Dec; 15(12):2287-94. [↩]
- World Cancer Research Fund; American Institute for Cancer Research. Food, nutrition, physical activity, and the prevention of cancer: a global perspective. Washington, DC: American Institute for Cancer Research; 2007. Nov. [↩]
- https://nutritionfacts.org/video/achieving-remission-of-crohns-disease/ [↩]
- Hou J.K., Abraham B., El-Serag H. Dietary intake and risk of developing inflammatory bowel disease: A systematic review of the literature. Am. J. Gastroenterol. 2011;106:563–573. doi: 10.1038/ajg.2011.44. [↩] [↩] [↩] [↩]
- Ananthakrishnan A.N., Khalili H., Konijeti G.G., Higuchi L.M., De Silva P., Korzenik J.R., Chan A.T. A prospective study of long-term intake of dietary fiber and risk of Crohn’s disease and ulcerative colitis. Gastroenterology. 2013;145:970–977. doi: 10.1053/j.gastro.2013.07.050. [↩]
- Racine A., Carbonnel F., Chan S.S.M., Hart A.R., Bueno-de-Mesquita H.B., Oldenburg B., Key T. Dietary patterns and risk of inflammatory bowel disease in Europe. Inflamm. Bowel Dis. 2016;22:345–354. doi: 10.1097/MIB.0000000000000638. [↩]
- Sakamoto N., Kono S., Wakai K., Fukuda Y., Satomi M., Shimoyama T., Kobashi G. Dietary risk factors for inflammatory bowel disease: A multicenter case-control study in Japan. Inflamm. Bowel Dis. 2005;11:154–163. doi: 10.1097/00054725-200502000-00009. [↩] [↩]
- Colitis (Severe), Inflammatory Bowel Disease, Ulcerative Colitis, Crohn’s Disease [↩] [↩]
- Jantchou P., Morois S., Clavel-Chapelon F., Boutron-Ruault M.-C., Carbonnel F. Animal protein intake and risk of inflammatory bowel disease: The E3N prospective study. Am. J. Gastroenterol. 2010;105:2195–2201. doi: 10.1038/ajg.2010.192. [↩]
- Andersson, H. Fat-reduced diet in the symptomatic treatment of small bowel disease. Metabolic studies in patients with Crohn’s disease and in other patients subjected to ileal resection. Gut 15:351, 1974 [↩]
- Reif S., Klein I., Lubin F., Farbstein M., Hallak A., Gilat T. Pre-illness dietary factors in inflammatory bowel disease. Gut. 1997;40:754–760. doi: 10.1136/gut.40.6.754. [↩]
- Diet as a risk factor for the development of ulcerative colitis. Geerling BJ, Dagnelie PC, Badart-Smook A, Russel MG, Stockbrügger RW, Brummer RJ. Am J Gastroenterol. 2000 Apr; 95(4):1008-13. [↩]
- “Digestive Health” from The Power of Food: Session 6 by Dr Neal Barnard [↩] [↩]
- Tjonneland A., Overvad K., Bergmann M.M., Nagel G., Linseisen J., Hallmans G., Palmqvist R., Sjodin H., Hagglund G., Berglund G., et al. Linoleic acid, a dietary n-6 polyunsaturated fatty acid, and the aetiology of ulcerative colitis: A nested case-control study within a European prospective cohort study. Gut. 2009;58:1606–1611. [↩]
- Fast Food Genocide: How Processed Food Is Killing Us and What We Can Do about It17 Oct 2017 by Fuhrman MD, Dr Joel [↩]
- Inflamm Bowel Dis. 2002 Sep;8(5):340-6. Potato glycoalkaloids adversely affect intestinal permeability and aggravate inflammatory bowel disease. Patel B, Schutte R, Sporns P, Doyle J, Jewel L, Fedorak RN. [↩]
- Feagan B.G., Sandborn W.J., Mittmann U., Bar-Meir S., D’Haens G., Bradette M., Hébuterne X. Omega-3 free fatty acids for the maintenance of remission in Crohn disease: The EPIC Randomized Controlled Trials. JAMA. 2008;299:1690–1697. doi: 10.1001/jama.299.14.1690. [↩]
- Turner D., Shah P.S., Steinhart A.H., Zlotkin S., Griffiths A.M. Maintenance of remission in inflammatory bowel disease using omega-3 fatty acids (fish oil): A systematic review and meta-analyses. Inflamm. Bowel Dis. 2011;17:336–345. doi: 10.1002/ibd.21374. [↩]
- Cabré E., Domènech E. Impact of environmental and dietary factors on the course of inflammatory bowel disease. World J. Gastroenterol. 2012;18:3814–3822. doi: 10.3748/wjg.v18.i29.3814. [↩]
- Cabré E, Mañosa M, Gassull MA. Omega-3 fatty acids and inflammatory bowel diseases – a systematic review. Br J Nutr. 2012;107(Suppl 2):S240–S252. [↩]
- Lev-Tzion R., Griffiths A.M., Leder O., Turner D. Omega 3 fatty acids (fish oil) for maintenance of remission in Crohn’s disease. Cochrane Database Syst. Rev. 2014;2:CD006320. [↩]
- Ananthakrishnan A.N., Khalili H., Konijeti G.G., Higuchi L.M., de Silva P., Fuchs C.S., Chan A.T. Long-term intake of dietary fat and risk of ulcerative colitis and Crohn’s disease. Gut. 2014;63:776–784. doi: 10.1136/gutjnl-2013-305304. [↩]
- Amre D.K., D’Souza S., Morgan K., Seidman G., Lambrette P., Grimard G., Chotard V. Imbalances in dietary consumption of fatty acids, vegetables, and fruits are associated with risk for crohn’s disease in children. Am. J. Gastroenterol. 2007;102:2016–2025. doi: 10.1111/j.1572-0241.2007.01411.x. [↩] [↩]
- John S., Luben R., Shrestha S.S., Welch A., Khaw K.-T., Hart A.R. Dietary n-3 polyunsaturated fatty acids and the aetiology of ulcerative colitis: A UK prospective cohort study. Eur. J. Gastroenterol. Hepatol. 2010;22:602–606. doi: 10.1097/MEG.0b013e3283352d05. [↩]
- But I thought Fish Was Good For Me! [↩]
- Uchiyama K., Nakamura M., Odahara S., Koido S., Katahira K., Shiraishi H., Tajiri H. N-3 polyunsaturated fatty acid diet therapy for patients with inflammatory bowel disease. Inflamm. Bowel Dis. 2010;16:1696–1707. doi: 10.1002/ibd.21251. [↩]
- Diet in the aetiology of ulcerative colitis: a European prospective cohort study. Hart AR, Luben R, Olsen A, Tjonneland A, Linseisen J, Nagel G, Berglund G, Lindgren S, Grip O, Key T, Appleby P, Bergmann MM, Boeing H, Hallmans G, Danielsson A, Palmqvist R, Sjodin H, Hagglund G, Overvad K, Palli D, Masala G, Riboli E, Kennedy H, Welch A, Khaw KT, Day N, Bingham S. Digestion. 2008; 77(1):57-64. [↩]
- Ananthakrishnan A.N., Khalili H., Konijeti G.G., Higuchi L.M., de Silva P., Fuchs C.S., Chan A.T. Long-term intake of dietary fat and risk of ulcerative colitis and Crohn’s disease. Gut. 2014;63:776–784. doi: 10.1136/gutjnl-2013-305304. [↩]
- Diet and inflammatory bowel disease: a case-control study. Persson PG, Ahlbom A, Hellers G. Epidemiology. 1992 Jan; 3(1):47-52. [↩] [↩]
- Niewiadomski O., Studd C., Wilson J., Williams J., Hair C., Knight R., Dowling D. Influence of food and lifestyle on the risk of developing inflammatory bowel disease. Intern. Med. J. 2016;46:669–676. doi: 10.1111/imj.13094. [↩]
- Opstelten J.L., Leenders M., Dik V.K., Chan S.S.M., van Schaik F.D.M., Khaw K.-T., Grip O. Dairy Products, Dietary Calcium, and Risk of Inflammatory Bowel Disease: Results From a European Prospective Cohort Investigation. Inflamm. Bowel Dis. 2016;22:1403–1411. doi: 10.1097/MIB.0000000000000798. [↩]
- Click RE, Van Kampen CL. Short communication: progression of Johne’s disease curtailed by a probiotic. J Dairy Sci. 2009;92:4846–4851. doi: 10.3168/jds.2009-2129. [↩]
- Dietary Treatment of Crohn’s Disease [↩]
- Truelove, S. Ulcerative colitis provoked by milk. Br Med J 1:154, 1961 [↩]
- Wright, R. A controlled therapeutic trial of various diets in ulcerative colitis. Br Med J 2:138, 1965 [↩]
- Is Nutritional Yeast Healthy for Everyone? Michael Greger M.D. FACLM September 4th, 2017 Volume 37. [↩] [↩]
- Hermon-Taylor J, Bull T. Crohn’s disease caused by Mycobacterium avium subspecies paratuberculosis: a public health tragedy whose resolution is long overdue. J Med Microbiol. 2002;51:3–6. [↩]
- Gut Pathog. 2010; 2: 21. Published online 2010 Dec 17. doi: 10.1186/1757-4749-2-21. Ulcerative colitis and Crohn’s disease: is Mycobacterium avium subspecies paratuberculosis the common villain? Ellen S Pierce. [↩]
- Scanu AM, Bull TJ, Cannas S, Sanderson JD, Sechi LA, Dettori G, Zanetti S, Hermon-Taylor J. Mycobacterium avium subspecies paratuberculosis infection in cases of irritable bowel syndrome and comparison with Crohn’s disease and Johne’s disease: common neural and immune pathogenicities. J Clin Microbiol. 2007;45:3883–3890. doi: 10.1128/JCM.01371-07. [↩]
- Olbe L. Concept of Crohn’s disease being conditioned by four main components, and irritable bowel syndrome being an incomplete Crohn’s disease. Scand J Gastroenterol. 2008;43:234–241. doi: 10.1080/00365520701676971. [↩]
- Expert Rev Gastroenterol Hepatol. Author manuscript; available in PMC 2016 Oct 16. Published in final edited form as: Expert Rev Gastroenterol Hepatol. 2015; 9(12): 1523–1534. Published online 2015 Oct 16. doi: 10.1586/17474124.2015.1093931. PMCID: PMC4894645. Mycobacterium paratuberculosis as a cause of Crohn’s disease. Adrienne L. McNees, Ph.D., Diane Markesich, Ph.D., Najah R. Zayyani, M.D., and David Y. Graham, M.D. [↩]
- Heaton, K. Treatment of Crohn’s disease with an unrefined-carbohydrate, fibre-rich diet. Br Med J 2:764, 1979 [↩]
- Joel Fuhrman on Frankenfoods: Fast Food Genocide. [↩]
- Butyrate – Why Dietary Fibre is So Important [↩] [↩]
- Chiba M, Tsuda H, Abe T, Sugawara T, Morikawa Y. Missing environmental factor in inflammatory bowel disease: diet-associated gut microflora. Inflamm Bowel Dis. 2011 Aug;17(8):E82–3. [↩]
- Zoetendal EG, de Vos WM. Effect of diet on the intestinal microbiota and its activity. Curr Opin Gastroenterol. 2014 Mar;30(2):189–95. [↩]
- Asakura H., Suzuki K., Kitahora T., Morizane T. Is there a link between food and intestinal microbes and the occurrence of Crohn’s disease and ulcerative colitis? J. Gastroenterol. Hepatol. 2008;23:1794–1801. doi: 10.1111/j.1440-1746.2008.05681.x. [↩]
- Packey C.D., Sartor R.B. Commensal bacteria, traditional and opportunistic pathogens, dysbiosis and bacterial killing in inflammatory bowel diseases. Curr. Opin. Infect. Dis. 2009;22:292–301. doi: 10.1097/QCO.0b013e32832a8a5d. [↩]
- Sartor R.B. Microbial Influences in Inflammatory Bowel Diseases. Gastroenterology. 2008;134:577–594. doi: 10.1053/j.gastro.2007.11.059. [↩]
- Zhang Y.-J., Li S., Gan R.-Y., Zhou T., Xu D.-P., Li H.-B. Impacts of gut bacteria on human health and diseases. Int. J. Mol. Sci. 2015;16:7493–7519. doi: 10.3390/ijms16047493. [↩]
- Curing Leaky Gut Syndrome [↩]
- Boosting Gut Flora without Probiotics [↩]
- “Leaky Gut” – Is it real and what to do about it? [↩]
- Ulcerative Colitis, Crohn’s Disease and Leaky Gut Syndrome with Michael Klaper, M.D. [↩]
- Barclay GR, McKenzie H, Pennington J, Parratt D, Pennington CR. The effect of dietary yeast on the activity of stable chronic Crohn’s disease. Scand J Gastroenterol. 1992;27(3):196-200. [↩]
- Does Nutritional Yeast Trigger Crohn’s Disease? Michael Greger M.D. FACLM August 30th, 2017 Volume 37 [↩]
- Gut mucosal response to food antigens in Crohn’s disease. J. VAN DEN BOGAERDE, J. CAHILL, A. V. EMMANUEL, C. J. VAIZEY, I. C. TALBOT, S. C. KNIGHT & M. A. KAMM. St Mark’s Hospital, Harrow, Middlesex, UK and Imperial College School of Medicine, London, UK. Accepted for publication 17 July 2002. [↩]
- Franzini L, Ardigò D, Valtueña S, Pellegrini N, Del Rio D, Bianchi MA, Scazzina F, Piatti PM, Brighenti F, Zavaroni I. Food selection based on high total antioxidant capacity improves endothelial function in a low cardiovascular risk population. Nutr Metab Cardiovasc Dis. 2010 Jul 29. [↩]
- Citrus. Topic summary contributed by volunteer(s): Paul [↩]
- Anti-Inflammatory Antioxidants. Michael Greger M.D. FACLM January 27th, 2012 Volume 7 [↩]
- Inflammatory Remarks about Arachidonic Acid. Michael Greger M.D. FACLM September 30th, 2011 Volume 5 [↩]
- Treating Crohn’s Disease With Diet. Written By Michael Greger M.D. FACLM on September 13th, 2012. [↩]
- National Cancer Institute Division of Cancer Control & Population Sciences. Epidemiology and Genomics Research Program [↩]
- https://academic.oup.com/ije/article/44/6/1995/2572627 [↩]
- Yu LC. Intestinal Epithelial Barrier Dysfunction in Food Hypersensitivity. J Allergy (Cairo) 2012;2012:96081. [↩]
- Chen BW Wang HH Liu JX Liu XG . Zinc sulphate solution enema decreases inflammation in experimental colitis in rats . J Gastroeneterol Hepatol 1999 ; 14 : 1088 – 92 . [↩]
- Iwaya H Kashiwaya M Shinoki A et al. . Marginal zinc deficiency exacerbates experimental colitis induced by dextran sulfate sodium in rats . J Nutr 2011 ; 141 : 1077 – 82 . [↩]
- Sturniolo GC Di Leo V Ferronato A D’Odorico A D’Inca R . Zinc supplementation tightens ‘leaky gut’ in Crohn’s disease . Inflamm Bowel Dis 2001 ; 7 : 94 – 98 . [↩]
- Lahiri A Abraham C . Activation of Pattern Recognition Receptors Up-Regulates Metallothioneins, Thereby Increasing Intracellular Accumulation of Zinc, Autophagy, and Bacterial Clearance by Macrophages . Gastroenterology 2014 ; 147 : 835 – 46 .)) ((Zhang X Giovannucci EL Smith-Warner SA et al. . A prospective study of intakes of zinc and heme iron and colorectal cancer risk in men and women . Cancer Causes Control 2011 ; 22 : 1627 – 37 . [↩]
- Bao S Liu MJ Lee B et al. . Zinc modulates the innate immune response in vivo to polymicrobial sepsis through regulation of NF-kappaB . Am J Physiol Lung Cell Mol Pysiol 2010 ; 298 : L744 – 54 . [↩]
- Finamore A Massimi M Conti Devirgiliis L Mengheri E . Zinc deficiency induces membrane barrier damage and increases neutrophil transmigration in Caco-2 cells . J Nutr 2008 ; 138 : 1664 – 70 . [↩]
- Prasad AS . Zinc: role in immunity, oxidative stress and chronic inflammation . Curr Opin Clin Nutr Metab Care 2009 ; 12 : 646 – 52 .)) ((Prasad AS Bao B Beck FW Sarkar FH . Zinc enhances the expression of interleukin-2 and interleukin-2 receptors in HUT-78 cells by way of NF-kappaB activation . J Lab Clin Med 2002 ; 140 : 272 – 89 . [↩]
- Prasad AS Bao B Beck FW Sarkar FH . Zinc-suppressed inflammatory cytokines by induction of A20-mediated inhibition of nuclear factor-kappaB . Nutrition 2011 ; 27 : 816 – 23 . [↩]
- Liu MJ Bao S Galvez-Peralta M et al. . ZIP8 regulates host defense through zinc-mediated inhibition of NF-kappaB . Cell Rep 2013 ; 3 : 386 – 400 . [↩]
- https://medical-dictionary.thefreedictionary.com/mineral+deficiency [↩]
- Digestion. 2010;81(4):252-64. doi: 10.1159/000264649. Epub 2010 Jan 30. Clinical relevance of IgG antibodies against food antigens in Crohn’s disease: a double-blind cross-over diet intervention study. Bentz S1, Hausmann M, Piberger H, Kellermeier S, Paul S, Held L, Falk W, Obermeier F, Fried M, Schölmerich J, Rogler G. [↩]
- BMC Gastroenterol. 2016; 16: 39. Published online 2016 Mar 22. doi: 10.1186/s12876-016-0450-3. Food IgG4 antibodies are elevated not only in children with wheat allergy but also in children with gastrointestinal diseases. Grażyna Czaja-Bulsa, corresponding author Michał Bulsa, and Aneta Gębala. [↩]
- Gunasekeera V, Mendall MA, Chan D, Kumar D. Treatment of Crohn’s Disease with an IgG4-Guided Exclusion Diet: A Randomized Controlled Trial. Dig Dis Sci. 2016 Apr;61(4):1148-57 [↩]
- Hidvegi E, Cserhati E, Kereki E, Savilahti E, Arato A. Serum immunoglobulin E, IgA, and IgG antibodies to different cow’s milk proteins in children with cow’s milk allergy: association with prognosis and clinical manifestations. Pediatr Allergy Immunol. 2002;13:255–61. doi: 10.1034/j.1399-3038.2002.01045.x. [↩]
- Savilahti EM, Savilahti E. Development of natural tolerance and induced desensitization in cow’s milk allergy. Pediatr Allergy Immunol. 2013;24:114–21. doi: 10.1111/pai.12004. [↩]
- Lee JH, Kim WS, Kim H, Hahn YS. Increased cow’s milk protein-specific IgG4 levels after oral desensitization in 7- to 12-month-old infants. Ann Allergy Asthma Immunol. 2013;111:523–8. doi: 10.1016/j.anai.2013.09.001. [↩]
- Vance GH, Grimshaw KE, Briggs R, Lewis SA, Mullee MA, Thornton CA, Warner JO. Serum ovalbumin – specific immunoglobulin G responses during pregnancy reflect maternal intake of dietary egg and relate to the development of allergy in early infancy. Clin Exp Allergy. 2004;34:1855–61. [↩]
- Ahrens B, de Oliveira LC L, Schulz G, Borres MP, Niggemann B, Wahn U, Beyer K. The role of hen’s egg-specific IgE, IgG and IgG4 in the diagnostic procedure of hen’s egg allergy. Allergy. 2010;65:1554–7. [↩]
- J Neurogastroenterol Motil. 2017 Oct; 23(4): 578–584. Published online 2017 Oct 30. doi: 10.5056/jnm17054. PMCID: PMC5628991. PMID: 28992678. Alterations of Food-specific Serum IgG4 Titers to Common Food Antigens in Patients With Irritable Bowel Syndrome. Hong Sub Lee1,2 and Kwang Jae Lee. [↩]
- World J Gastrointest Pathophysiol. 2016 Aug 15; 7(3): 276–282. Published online 2016 Aug 15. doi: 10.4291/wjgp.v7.i3.276. Pancreatic disorders in inflammatory bowel disease. Filippo Antonini, Raffaele Pezzilli, Lucia Angelelli, and Giampiero Macarri. [↩]
- J Clin Gastroenterol. 2010 Apr;44(4):246-53. doi: 10.1097/MCG.0b013e3181cadbe1. Pancreatitis in inflammatory bowel diseases. Pitchumoni CS1, Rubin A, Das K. [↩]
- Peppermint Oil for Irritable Bowel Syndrome. Michael Greger M.D. FACLM July 10th, 2015 Volume 25. [↩]
- Br J Pharmacol. 2017 Jun;174(11):1426-1449. doi: 10.1111/bph.13632. Epub 2016 Oct 25. Probiotics, fibre and herbal medicinal products for functional and inflammatory bowel disorders. Currò D1, Ianiro G2, Pecere S2, Bibbò S3, Cammarota G. [↩]
- Curcumin as “Curecumin”: from kitchen to clinic. Goel A, Kunnumakkara AB, Aggarwal BB. Biochem Pharmacol. 2008 Feb 15; 75(4):787-809. [↩]
- Hanai H., Iida T., Takeuchi K., Watanabe F., Maruyama Y., Andoh A., Yamada M. Curcumin maintenance therapy for ulcerative colitis: Randomized, multicenter, double-blind, placebo-controlled trial. Clin. Gastroenterol. Hepatol. 2006;4:1502–1506. doi: 10.1016/j.cgh.2006.08.008. [↩]
- Taylor R.A., Leonard M.C. Curcumin for inflammatory bowel disease: A review of human studies. Altern. Med. Rev. 2011;16:152. [↩]
- Kumar S., Ahuja V., Sankar M.J., Kumar A., Moss A.C. Curcumin for maintenance of remission in ulcerative colitis. Cochrane Database Syst. Rev. 2012 doi: 10.1002/14651858.CD008424.pub2. [↩]
- Turmeric, article by Dr Michael Greger [↩]
- Intest Res. 2017 Jan; 15(1): 103–108. Published online 2017 Jan 31. doi: 10.5217/ir.2017.15.1.103. Status of serum vitamin B12 and folate in patients with inflammatory bowel disease in China. Shaozhong Huang, Jiayi Ma, Mingming Zhu, and Zhihua Ran. [↩]
- Bermejo F, Algaba A, Guerra I, et al. Should we monitor vitamin B12 and folate levels in Crohn’s disease patients? Scand J Gastroenterol. 2013;48:1272–1277. [↩]
- USDA Article: B12 Deficiency May Be More Widespread Than Thought. By Judy McBride. August 2, 2000. [↩]
- Inflamm Bowel Dis. 2006 Mar;12(3):185-91. Nutritional deficiencies in patients with Crohn’s disease in remission. Filippi J, Al-Jaouni R, Wiroth JB, Hébuterne X, Schneider SM. (( JPEN J Parenter Enteral Nutr. 2007 Jul-Aug;31(4):311-9. Nutrition assessment of patients with inflammatory bowel disease. Vagianos K1, Bector S, McConnell J, Bernstein CN. [↩]
- Inflamm Bowel Dis. 2012 Oct;18(10):1961-81. doi: 10.1002/ibd.22906. Epub 2012 Apr 5. Micronutrient deficiencies in inflammatory bowel disease: from A to zinc. Hwang C1, Ross V, Mahadevan U. [↩]
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5847182/ [↩]
- Ogura Y, Bonen DK, Inohara N, et al: A frameshift mutation in NOD2 is associated with susceptibility to Crohn’s disease. Nature 2001;411:603-606. [↩]
- Gastroenterol Hepatol (N Y). 2013 Nov; 9(11): 752–755. PMCID: PMC3995201. PMID: 24764796. Isotretinoin, Acne, and Crohn’s Disease: A Convergence of Bad Skin, Bad Science, and Bad Litigation Creates the Perfect Storm. [↩]